Demand for Prescription Drugs:
The Effects of Managed Care Pharmacy Benefits
Rika Onishi Mortimer
University of California, Berkeley
Department of Economics
549 Evans Hall, #3880
Berkeley, CA 94720-3880
November 12, 1997
Abstract
This paper examines how demand for prescription drugs is influenced by different types of insurance. In order to understand demand characteristics and the competitiveness of pharmaceutical markets, both intermolecular (therapeutic) and intramolecular (generic) substitutions are studied in the antidepressant and beta blocker (anti-hypertensive) markets. Mixed logit and other discrete choice models are applied to national survey and product sales data. The results indicate that demand in managed care sectors is more price elastic than in other sectors. Demand in the self-paid sector is found to be the least price elastic, despite the fact that patients must pay for the entire cost of drugs. The results confirm the effectiveness of managed care incentives in shifting prescription patterns toward less expensive products, and suggest the existence of an agency problem between physicians and patients.
I would like to thank IMS America for generously providing data for this research. I am also grateful to Bronwyn Hall, Theodore Keeler, Kenneth Train, Richard Scheffler, Scott Stern and Sean Sullivan for their valuable comments and advice. I am also indebted to Erik Heitfield, Jimmy Chan, John Giles, Daniel Covitz and Jim Prieger for helpful suggestions. Finally, I would like to thank my husband Scott for his support and patience during my dissertation writing. All mistakes are my own.
During the 1980s, the producer price index for prescription drugs more than doubled, significantly outpacing the average producer price index (Standard & Poor, 1995). The pharmaceutical industry also experienced outstanding earnings growth, averaging eighteen percent per year during the period from 1982 to 1992 (Standard & Poor, 1993). The inflation of drug prices and high profitability can be attributed to many factors. One contributor is strong patent protection. Many pharmaceutical manufacturers have had monopolies or near-monopolies in some key therapeutic markets, enabling companies to maintain and increase drug prices. Also, demand for drugs has historically been insensitive to price increases, due to third-party reimbursement. Since neither patients nor physicians paid for the cost of medication, the price of drugs was not the most important consideration in physicians’ choice of drugs.
Prescription drug markets have changed considerably in recent years, due to governmental scrutiny of the drug industry and the rapid growth of managed care. As a result of becoming a prime target of President Clinton’s healthcare reform plan in 1993, the pharmaceutical manufacturers were accused of making excessive profits from sick and elderly citizens. The healthcare reform initiatives included limits on drug prices and Medicaid reimbursement, as well as other regulatory controls. In order to respond to intense criticism from government and consumer groups and avoid possible regulation, in 1993 seventeen leading drug manufacturers volunteered to limit their annual price increases to the consumer price index in 1993. Although the failure of healthcare reform initiatives in 1994 relieved some pressure on drug manufacturers, heightened publicity on the pharmaceutical industry raised public awareness of drug prices. Also, the emergence of managed care as a major player in the prescription marketplace has recently altered the way drugs are prescribed. In the past, physicians primarily determined which drug to prescribe. In order to contain escalating prescription drug costs, managed care providers employ various methods to control and influence physicians’ prescription decisions. For example, managed care plans and Medicaid programs implement formularies (regulated lists of approved medications) to restrict reimbursement to a fairly small number of cost-effective drugs in a given therapeutic class, thus encouraging the utilization of less expensive drugs over more expensive ones (often referred to as "therapeutic substitution"). Also, managed care plans often encourage the use of generic drugs whenever applicable (often referred as to "generic substitution"), since generic drugs are the chemical and therapeutic equivalents to brand name drugs. The effective managed care measures foster higher price elasticity of demand, more competitive prescription drug markets, and slower drug price increases. As the managed care sector grows in the future, the pharmaceutical industry may be unlikely to maintain the high profit growth and price increases similar to those achieved in the 1980s to early 1990s.
The growth of the managed care sector has been remarkable: it accounted for about twenty percent of total pharmaceutical purchases in 1990 and reached roughly fifty percent in 1995 with growth expected to continue (Standard & Poor, 1995). In order to understand the implications of changes in the pharmaceutical industry due to the movement toward markets dominated by managed care, it is important first to study characteristics of prescription drug markets. Specifically, it is crucial to understand differences in the degree of price sensitivity and the degree of perceived product differentiation in the drug markets. This research attempts to answer the question by examining how demand for prescription drugs is influenced by different types of health insurance. Patients with drug coverage are more likely to purchase expensive drugs than patients who have to pay for the entire cost of drugs themselves. Managed-care patients are more likely to be prescribed a less expensive alternative among drugs which are believed to have similar efficacy. The own- and cross-price elasticity of a drug and the degree of substitutability among similar drugs are, therefore, affected by the patients’ coverage status as well as by the restrictions placed on coverage by health insurance policies.
In order to measure the degree of perceived product differentiation among brand name products and between brand name and generic products as a function of the type of insurance patients have, multinomial logit, nested logit, and mixed logit (random parameters logit) models are estimated. These models all produce demand forecasts with an assumption that a physician chooses a drug she considers to have the maximum net benefit to her patient, given that the patient needs to be prescribed a drug from among alternatives with similar therapeutic effects. The differences among the logit models come from the assumption imposed on the distribution of unobserved error terms associated with the utility from each alternative. The mixed logit model allows the most flexible distributions of error terms and seems to be the best model to describe the demand structure of prescription drugs.
The data used in this study are the National Ambulatory Medical Care Survey (NAMCS) 1991-1993 and pharmaceutical marketing and sales data supplied by IMS America, a market research firm in Pennsylvania. The NAMCS consists of a national sample of patients using ambulatory medical care services, reporting information on patient office visits such as patient characteristics, insurance status, physician characteristics, and the name of medications prescribed. The IMS data, on the other hand, contain retail drug prices and advertising costs of each drug. One can expect that patient and physician characteristics as well as drug characteristics influence demand for drugs. Two different therapeutic classes of medicine are studied in order to find the effects of patient and physician characteristics on prescription patterns, which may be common or different across various therapeutic markets. The two classes I consider are antidepressants and beta blockers (anti-hypertensive drugs).
Because of cost-cutting measures such as formularies and more controlled health care management often seen in staff-model HMOs, it is expected that Medicaid and HMO sectors have higher own-price elasticities and cross-price elasticities among brand name drugs and between generics and brand name drugs than other insurance sectors. My results confirm the effectiveness of the cost-cutting measures of the managed care sectors. For patients without any health insurance, there are two forces determining their demand characteristics. Since they have to pay out of their pockets for the entire cost of drugs (i.e., the absence of moral hazard), the self-paid sector may have more elastic demand and greater substitutability among alternative drugs than the insured sector. On the other hand, due to their insufficient knowledge of available alternative drugs and their physician’s possible lack of concern for their expenses (i.e., an agency problem), the self-paid sector may exhibit less elastic demand and lower substitutability. My results show that the latter force dominates the former, suggesting that a physician may not be a perfect agent for her patients. Therefore, adopting sophisticated drug-cost management plans seems to be more effective in reducing rising prescription drug costs than making patients pay for the entire cost of drugs.
The outline of this paper is as follows. The next section reviews the literature. Section 3 discusses the structure of prescription drug markets and issues regarding managed care and the pharmaceutical industry. Section 4 describes the data used in this research. Section 5 presents a model of demand for pharmaceutical products and empirical specifications. Results for the antidepressant and beta blocker markets are discussed in Section 6. Finally, Section 7 draws conclusions.
Several empirical studies on prices of prescription pharmaceutical products have examined the relationship between brand name products and generic products and investigated price changes of brand name products due to their patent expiration (Grabowski and Vernon, 1992; Caves, Whinston, and Hurwitz, 1991; Frank and Salkever, 1994; Wiggins and Maness, 1994). Although the findings of these studies are somewhat mixed, there seems to be general consensus that prices of branded products do not decline significantly after generic entry. It appears that pharmaceutical companies often find it more profitable to target customers with high willingness-to-pay and high brand name loyalty than to engage in fierce price competition against generics. These studies focus on competition between brand name drugs and generics of the same chemical compound (i.e., intramolecular competition - Ibuprofen vs. Motrin) but exclude competition between differentiated products that do not share the same molecule (i.e., intermolecular competition - Motrin vs. Aleve). In general, difficulty in defining markets with different molecular entities and complexity in comparing the quality differences of various molecules make analysis involving therapeutic competition very complex. Two recent studies by Stern (1994) and Ellison, Cockburn, Griliches and Hausman (1997) explore the substitutability among different molecules and between brand name drugs and generics.
Stern’s work (1994) is one of the first attempts to incorporate both generic substitution and therapeutic substitution. Stern estimates demand for pharmaceutical products and parameterizes the degree of substitutability between differentiated products in narrowly defined therapeutic markets: gout therapies, non-barbituate sedatives, oral diabetic therapies, and minor tranquilizers. The application of a two-level nested logit model to product-level data allows clustering products into mutually exclusive molecular groups and generic/brand name groups so that the substitutability among molecules and between pioneer and generic products can be estimated. The principal finding of the study is that alternative pioneer products (i.e., the oldest branded product of each molecule) are relatively strong substitutes for each other, whereas pioneer products are differentiated from generic products of the same molecule in three of the four markets analyzed.
The result that differentiated brand name products are fairly close substitutes is interesting, because it suggests that the market power of patented products may not be as great as one might think. However, the result may be sensitive to different definitions of markets. It is often difficult to group products into various therapeutic classes, since many drugs have multiple indications. It seems that Stern defines therapeutic markets based on IMS America’s product classifications and focuses on narrowly defined therapeutic classes. Therefore, it is important to evaluate the robustness of his results with more widely defined markets and determine whether or not the finding of high therapeutic substitutability was driven by the narrow definition of markets.
Ellison, Cockburn, Griliches and Hausman (1997) explore demand characteristics of four Cephalosporins (antibiotics) and study both generic and therapeutic substitutions. They model demand as a multistage budgeting problem, in which the prescribing stage is the choice of molecule and the dispensing stage is the choice between brand name drugs and generics. From monthly wholesale sales data from 1985 to 1991 supplied by IMS America, "conditional" elasticities (i.e., elasticities conditional on a particular molecule and defined for branded and generic products of the same molecule) and "unconditional" elasticities are computed. Their main conclusion is that demand for a branded (generic) product appears to be responsive to a price change of its generic (branded) equivalent, but is not always responsive to a price change of its therapeutic substitutes. This result contradicts Stern’s findings of the low substitutability between a branded and a generic drug of the same molecule and high substitutability between different molecules. They also find that generic drugs are more price elastic than their branded equivalents.
Although the sequential decision model employed by Ellison et al. is a reasonable generalization of pharmaceutical demand, it could be an oversimplification of the process by which a particular drug is chosen. Since 1989, all states have had laws allowing generic substitution by pharmacists unless physicians specifically prohibit substitution on dispensing of their prescriptions (Health Care Financing Administration, 1993). Thus, one can argue that the choice of generics vs. branded drugs arises at the dispensing level. However, physicians sometimes prohibit generic substitution and also specifically note a generic name on their prescription pads. If such situations often arise, characterizing pharmaceutical demand as the two-stage decisions by physicians and pharmacists is not very appealing. How often physicians make a decision on the choice of a product (e.g., branded or generic products) in addition to the choice of a molecule greatly depends on patients’ insurance policies. Some plans have the policy of mandatory generic substitution at the dispensing level and others provide physicians incentives to choose generics. My study provides some answers to this question: generic substitution policies often employed in HMO and Medicaid plans affect physicians’ prescription behavior differently for HMO and Medicaid patients. It appears that HMO patients are more likely to receive generically written prescriptions than other patients, ceteris paribus. This finding is consistent with the result of another demand study by Hellerstein (1994).
Hellerstein (1994) investigates the question of why brand name drugs are often prescribed instead of less expensive generic drugs. She incorporates the effects of insurance, but the focus of the study is on competition between branded and generic products and not between therapeutic equivalents. Using the 1989 National Ambulatory Medical Care Survey, the effects of physician and patient characteristics on prescription decisions between branded and generic products are estimated. Hellerstein also evaluates the effects of state legislation such as mandatory generic substitution on physician’s prescription patterns. The 1989 NAMCS data consist of more than 35,000 patient-visits to office-based physicians in the US. Each record includes a patient’s age, sex, race, insurance type, physician’s specialty, location and diagnosis, and the name of medication prescribed. The estimation of binomial probit models, where the choice is either generics or brand name drugs, reveals that women and older people are less likely to be prescribed generics. Also, Hellerstein finds that the effects of mandatory substitution laws on prescription patterns are insignificant. One interesting result is the lack of strong evidence that the probability of generic prescription varies systematically with the patient insurance status, except that HMO patients are more likely to receive generics (due to HMO physicians’ tendencies to prescribe generics).
A weakness of Hellerstein’s study is that neither prices nor any other product characteristics are included in her discrete choice estimation. This is due to the fact that the NAMCS does not contain any data regarding drug characteristics such as wholesale prices and the age of products. In order to accommodate the lack of data on drugs, therapeutic class specific dummy variables are used to allow separate intercepts for different therapeutic classes. These dummy variables are class specific but not drug specific, in order to reduce the dimensionality of estimation. This is problematic since Hellerstein’s therapeutic classification is large (e.g., cardiovascular, hormones, metabolics, etc.) and she assumes that the quality and price differentials between branded drugs and generics are all the same for drugs within the therapeutic class. It is reasonable to suspect that there are significant variations in the price and quality differentials between branded drugs and generics among widely defined drug classes. The therapeutic class dummies may be too aggregated to sufficiently capture the quality and price differential between branded and generic drugs. Also, evaluating different drug classes together makes it difficult to interpret the meaning of coefficients on age, sex, and race, except that they might reflect the effects of income status of patients.
Stern and Ellison et al. investigate both intermolecular and intramolecular competition, whereas Hellerstein evaluates the effects of insurance and other physician/patient characteristics on intramolecular competition. My analysis attempts to go further by incorporating the effects of insurance on intermolecular competition as well as intramolecular competition. In other words, I examine the degree of substitutability among the same molecules as well as between different molecules in therapeutic classes and evaluates how substitutability is affected by the patient’s insurance status.
The prescription drug market can be segmented into retail and non-retail markets. The retail market involves the distribution channel through retail pharmacies and the non-retail market involves other channels such as hospitals, the Veterans Administration, long-term care, military, etc. Mail-order systems are an additional category. This study focuses on the retail prescription drug markets.
For the retail market, pharmaceutical manufacturers sell to wholesalers and sometimes sell to large chain drug stores directly. Consumers in this market consist of patients with managed care pharmacy plans (e.g., plans offered by Medicaid, pharmacy benefit managers (PBMs) and HMOs) and unmanaged care patients (e.g., patients with indemnity plans or without drug coverage). Managed care pharmacy is a program in which health care providers or insurers attempt to control expenditures for prescription drugs by establishing and enforcing a formulary and other related services. The main goal of managed care pharmacy is to improve the outcome of pharmaceutical therapies while cost effectively managing the utilization of drugs. The next few sections describe how prescription drug consumption is controlled by various measures in managed care plans and summarize the prescription coverage of Medicaid patients. Since the Medicaid pharmacy policies changed during the sample data period in the analysis, it is important to understand what these changes are and how they may impact the demand analysis.
Health Maintenance Organizations and Pharmacy Benefit Managers
Many HMOs began to adopt restrictive formularies to manage their drug costs in the last fifteen years. HMOs sometimes develop their formularies independently by forming pharmaceutical and therapeutics (P&T) committees. The P&T committee, consisting of physicians and health care professionals, makes decisions regarding formularies and other treatment protocols. HMOs sometimes utilize subcontractors to monitor and control their drug expenditures. Since there are several similar compounds (including brand name and generic drugs) in many therapeutic classes, these organizations can choose compounds that are efficacious and cost effective for inclusion in their formulary. During 1991, forty-nine percent of HMOs used some type of formulary. Staff model HMOs were shown to be the most successful at controlling pharmaceutical costs (Merck, 1993). HMOs adopt various financial incentives to encourage doctors and affiliated pharmacists to prescribe and dispense generics and lower priced brand name drugs. Figure 1 shows the basis of financial incentives for controlling physicians’ prescribing patterns used by HMOs. Many HMOs monitor physicians’ prescribing patterns through pharmacy data and identify high prescribers of non-formulary or high-cost drugs. Those physicians who do not conform to prescription guidelines often receive education on cost-effective treatment alternatives and in some cases are subject to financial penalties.
Pharmaceutical benefit management companies (or pharmaceutical benefit managers --- often called PBMs) are firms that administer pharmaceutical benefit plans for insurers or employers who do not have the expertise in drug management or who find it more appropriate to purchase the services externally. HMOs may also subcontract with PBMs to manage their prescription drug benefits. PBMs arose in the 1970s and began to grow rapidly in the 1980s and the early 1990s, due to increases in expenditures for prescription drugs, rising employers/insurers’ concerns to contain health costs, and the growth of managed health care in general. The size and variety of PBMs’ services have expanded over the years and their services are expected to continue changing in the future. The traditional PBM role is that of a claim processor rather than of a pharmacy care manager. Today, PBMs control pharmaceutical expenses by establishing networks of pharmacy providers, negotiating rebates and discounts with manufacturers, developing drug formularies and pre-authorization systems, reviewing drug utilization services, and providing prescription mail-order services. On-line adjudication systems, which were added to PBM services in the late 1980s, verify patients’ eligibility at the point of service and perform drug utilization review (DUR). DUR is a study of a patient’s drug utilization, which provides subsequent recommendations to the patient’s physician and pharmacist regarding the most efficient and cost-effective drug treatment. DUR evaluates the extent of physician compliance with formularies and educates physicians about cost differentials between various treatments. It is reported that over ninety percent of community pharmacies are connected on-line to at least one third-party pharmaceutical claims processor today.
The Medicaid program provides medical assistance for certain individuals and families with low incomes and financial resources. It is administered under the guidance of the Health Care Financing Administration (HCFA), and each state manages its Medicaid program through its Medicaid agency. Since each state establishes its own eligibility standards and determines the type of services and the rate of payment, the Medicaid program varies from state to state. However, prescription drug coverage is provided by Medicaid programs in all states.
In 1987, the HCFA adopted a Federal maximum upper limit on payment for prescription drugs in order to curtail Medicaid drug expenditures. The reimbursement for multi-source drugs (those marketed or sold by multiple manufacturers or labelers) was set not to exceed 150% of the published price for the least costly therapeutic equivalent drug. For other drugs the limit was determined to be the lower of the estimated acquisition cost plus a reasonable dispensing fee or the pharmacist’s usual and customary charge to the public. In addition to the upper limit on reimbursement, many states adopted formularies to control Medicaid drug expenditures. In 1990, twenty states had restricted formularies that excluded many drugs from Medicaid reimbursement or specified drugs that require physicians to obtain prior approval. A study found that Medicaid restricted formularies reduce its drug expenditures.
The Omnibus Budget Reconciliation Act of 1990 (OBRA ’90) made significant changes to Medicaid pharmacy benefits. First, it mandated a rebate program where participating drug manufacturers rebate to the state Medicaid program the differences between the price charged to Medicaid and the prices below Medicaid levels charged to other customers. In return, the participating drug manufacturers obtained unrestricted access to Medicaid formularies. In short, the rebate program replaced formularies. Although state formularies were eliminated, Medicaid prescription drug utilization continued to be managed by other means. The upper limit on the reimbursement rate remained to encourage consumption of less expensive drugs. Some states enforced mandatory generic substitutions and DUR. In fact, OBRA ’90 mandated all states to perform DUR by January of 1993. Specifically, all states were required to conduct prospective and retrospective reviews for analyzing drug utilization patterns and to set up an education program for physicians and pharmacists. Also, OBRA ’90 required all states to cover all new drugs approved by the Federal Drug Administration (FDA) after January 1991 for a period of six months. At the end of six months, the state Medicaid programs were given the right to decide whether or not to restrict coverage of these new drugs through prior approval.
The Omnibus Budget Reconciliation Act of 1993 (OBRA ’93) repealed several provisions of OBRA ’90. OBRA ’93 reversed an OBRA ’90 mandate of the six-month period for new drugs in which no prior authorization was necessary. Also, forty percent of the U.S. returned to using formularies in 1993. Considering how short the period of suspension of formularies was, it is likely that physicians and pharmacists might have made no adjustment of their prescribing and dispensing behavior due to the elimination of formularies. In other words, physicians and pharmacists most likely formed habits of complying with state formularies and the effects of the temporary elimination of formularies were probably not very significant. The results shown in Section 6 are, in fact, consistent with this view, and the Medicaid sector seems to have been more price elastic than the less managed sectors during the period of 1991 to 1993. Figure 2 shows the percentage of states implementing Medicaid drug utilization controls in 1993.
Although the governmental regulatory pressure has been reduced due to the failure of healthcare reform and the dominance of industry-friendly Republicans in Congress, pharmaceutical manufacturers continue to face an increasingly cost-constrained marketplace. In 1994, the sales of third-party managed care prescriptions exceeded that of the cash pay sector for the first time. In 1995, more than fifty percent of total pharmaceutical retail sales were from the managed care sector, compared to about twenty percent in 1990 (Standard & Poor, 1995). Managed care pharmacy continues to cover more lives and improve techniques to control prescription patterns and it seems to have changed the market environment for pharmaceutical firms drastically. In this situation, it is important to understand the differences in demand characteristics in various segments of the markets grouped by insurance type and how the growth of managed care may impact the competitiveness of the prescription drug markets. As of today, there have not been many studies focusing on the impact of managed care pharmacy on the price competition of pharmaceutical firms. This paper attempts to evaluate the effects of managed care measures on the price elasticities of demand for prescription drugs.
Primary data used are from the National Ambulatory Medical Care Survey (NAMCS) 1991-1993. The NAMCS has been conducted annually since 1989 by the Division of Health Care Statistics of the National Center for Health Statistics, Centers for Disease Control and Prevention. The data consist of national samples of patient visits to the offices of nonfederally employed physicians, but exclude visits to hospital-based physicians and physicians primarily engaged in training, research, or administration. Therefore, outpatient markets are the focus of the survey. The NAMCS contains patient characteristics (e.g., age, sex, race, ethnicity, geography, insurance type), physician characteristics (e.g., specialty, geography), the patient’s reasons for visit, the physician’s diagnoses and treatment, and the choice of prescription. The NAMCS was designed to allow physicians to code up to five drugs prescribed to a patient per office visit, including both prescription and over-the-counter medications. There were 33,795 patient records provided by 1,354 doctors in 1991, 34,606 patient records by 1,558 doctors in 1992, and 35,978 patient records by 1,802 doctors in 1993.
The NAMCS specifies eight categories for expected source(s) of payment: HMO/other pre-paid, Medicare, Medicaid, other government, private/commercial, self-paid, no charge, and other. It should be noted that these are expected sources of payment for office visits and not necessarily for medication ordered. Therefore, it is not appropriate to directly apply this classification in estimating demand for prescription drugs as the type of prescription benefits. For example, standard Medicare benefits do not include prescription drug coverage and it is not possible to predict what type of drug insurance the patients with only Medicare coverage might have. A similar argument applies to categories such as no charge, other government, and other. Therefore, I focus only on commercial plans, HMO plans, Medicaid plans, and self-paid. By doing so, I make a reasonable assumption that patients with commercial health insurance plans also have drug benefits whereas patients with no health insurance do not. Furthermore, if a patient has multiple insurance plans, and it is impossible to infer which plan may cover the particular prescription in data, she is excluded from the analysis.
In addition to the fact that various average co-payment rates of each type of insurance affect demand for drugs differently, one can also predict that prescription patterns of Medicare and HMO patients differ from those of commercial and self-paid patients, due to both more use of and more successful adaptation of cost-cutting measures. Since the NAMCS does not provide information on the type of pharmacy benefit plans each patient has, it is not possible to identify patients with managed pharmacy benefits in the commercial sector. Some patients with a commercial health plan have card programs for prescription benefits offered by PBMs. Since PBMs implement formularies and drug utilization reviews, prescriptions for these patients are not "un-managed" in the sense of traditional indemnity plans. Therefore, the commercial category is not an equivalent term to the un-managed drug benefits category. This restriction in the data is expected to reduce the differences in price sensitivity between the HMO and commercial sectors.
Although the NAMCS provides data on prescription patterns, it does not include drug characteristics such as marketing costs and prices. Variables involving drug characteristics are mainly supplied by data from IMS America. IMS America provides vast collections of data on marketing and sales of the U.S. pharmaceutical industry. The IMS Office Promotion Report and National Journal Audit contain marketing data such as promotional expenditures to office-based physicians and spending on medical journal advertising of pharmaceutical products. These are appropriate measures for advertising cost for the outpatient market, which is a focus of the NAMCS data. In this research, these two figures are aggregated to construct one variable for marketing cost of a drug.
The IMS National Prescription Audit (NPA) provides average prices of prescription drugs at the retail level (i.e. the price that pharmacists charge), which were calculated from data consisting of a national sample of more than 20,000 stores including chain, independent and foodstore pharmacies. However, the price data are not broken down to the segments of the retail-outpatient markets. Therefore, the average retail price of each drug is not available for each insurance category defined in the NAMCS. In addition, prices of pharmacies that are restricted to serve only HMO patients (i.e. in-house pharmacies) are not in the scope of this audit. As a result, this restriction of price data, along with the constraint of NAMCS data where there is no information on the exact name of insurance plan of the patient, leads to using the same price data for all patients regardless of the patient’s insurance status. Although the price data are not specific to each insurance category, the average retail price is still a good indicator of price differentials of various drugs in all insurance categories. More costly drugs in one insurance category, say the cash payment sector, are also relatively expensive in other categories such as HMOs. Also, those drugs are more likely to be scrutinized by P&T committees developing formularies and treatment protocols. Therefore, the restriction of price data should not be detrimental to my analysis.
In order to compare prices of different drugs, the price of each drug is standardized to be the cost of average daily doses. The most desirable measure of price is an average cost to cure patients. However, such a measure is not widely available and there is no apparent consensus on such numbers for each drug. Given the absence of the desirable measure of price, the next best alternative is the average price per day. Since patients with depression or hypertension are often on medications continuously for a long period of time, the price per day seems to be more appropriate than the price of a prescription. As opposed to treatment with antibiotics, where one prescription often cures a malady, antidepressants and anti-hypertensive drugs are considered to be "maintenance" drugs. Therefore, the price that matters to patients and their insurance companies is the cost per day rather than the cost of a prescription. The costs of average daily doses are calculated from the average retail price of an extended unit (such as a tablet and a capsule) and the average quantity consumed provided in the IMS-NPA data.
Another characteristic of drugs is their age, which is the number of years that the product has been available to the public. The FDA approval date of each product is compared to the IMS product introduction date, the date when the product first appeared in the IMS data. Most products are launched in the market right after they obtain an approval form the FDA, but some do not become available until many months after approval. The delay may occur for many different reasons. For instance, marketing strategies such as label and packaging designs may not be finalized on time. Another example is the case in which the manufacturer of a branded product may obtain approval for a generic equivalent with the anticipation of marketing at the patent expiration of its branded drug. IPR Pharmaceuticals, which was the first to receive approval for their generic product, Atenolol (a beta blocker agent), did not launch their generic drug until 3 years after the approval date. Since IPR also owns Tenormin, the brand name version of Atenolol, the firm waited to market the generic version until a few months before the expiration of the patent for Tenormin. These examples show that the approval date and the product launch date do not necessarily match. Therefore, I computed the age of products mainly from the IMS product introduction date. If the IMS product introduction date and the FDA approval date differ by any significant amount, I checked with manufacturers to confirm the product introduction date.
Two therapeutic markets are studied in this research: antidepressants and beta blockers. These two markets are chosen mainly for two reasons. Due to their frequent usage, the number of observations of these drug classes is much greater than that of other classes of medication in the NAMCS data. Also, these two markets are relatively easily defined. The classification of products varies in the pharmacological literature, since many drugs have multiple indications. It appears that there exists reasonable consensus on the classification of these two markets, although the definition of many therapeutic classes is not always straightforward. After reviewing some pharmacological literature, including American Hospital Formularies, Drug Facts and Comparisons, and Drug Guide for Nurses, and comparing to the classification used by IMS, I have excluded molecules from a certain therapeutic class unless they are classified in the therapeutic class in most publications. Also, any product which is not entered in the FDA Orange Book is removed from my analysis.
The Data Appendix describes product characteristics and summary statistics for the antidepressant and beta blocker markets.
Suppose patient i sees physician j who makes a decision on the choice of prescription for patient i. Also, assume that the diagnosis for patient i determines a therapeutic class from which a prescription drug is selected. For now, assume that physician j is a perfect agent for patient i in terms of assessing the drug cost to patient i. That is, physician j takes patient i’s expenses into full consideration in choosing a drug. Suppose that the value of drug k which belongs to therapeutic class X to patient i is Vik. Assuming patient i has to pay for the full cost of drug k, physician j chooses drug k such that
Vik - Pk ![]() Vih - Ph for all k, hÎ
X .
Vih - Ph for all k, hÎ
X .
The value of drug k is determined by a patient’s characteristics (e.g., age, sex) as well as by the drug quality (e.g., tolerability level and efficacy). For example, in the case of heart disease, physicians generally perceive young patients to have lower risk than older patients. Accordingly, physicians may be more inclined to believe that the value of medication is lower for young patients than middle-aged or elderly patients. Therefore, the value of drug k has subscript i as well as k.
In general, the value of drug k to patient i is not perfectly observable at the time of prescription. More specifically, the expected value of drug k to patient i is affected by physician j’s perception of drug k, alternative drugs, and possible errors in evaluating the benefit of treatment for patient i. For instance, physician j may have a tendency to overvalue or undervalue certain drugs. Different physicians do not necessarily have the same opinions on the effectiveness of different drugs. Therefore, physician j chooses drug k such that
Vijk- Pk ![]() Vijh- Ph for all k, hÎ
X.
Vijh- Ph for all k, hÎ
X.
The expected value of drug k to patient i is denoted as Vijk, where subscript j reflects the fact that there exist physician specific effects on evaluation of treatment.
Suppose now patients have insurance coverage. Then, the cost of drug k to patient i is not the full price Pk. Assuming that physician j ignores the cost of drug k to a policy provider (due to the assumption that physician j is a perfect agent for patient i, i.e., she takes the cost of drugs to her patient into full consideration), the price considered in selecting drug k is gi(Pk) where gi(× ) is a function which maps Pk into the amount for which patient i pays. For the patients without coverage, gi(Pk) is obviously equal to Pk. Furthermore, it should be noted that the price of drug k varies depending on the insurance plan that patient i has. For example, the rate of reimbursement to pharmacists is sometimes negotiated between big drug chains and insurance companies and is often influenced by insurance companies and plan sponsors. Also, the reimbursement rate for Medicaid patients varies among states. Therefore, the price of drug k is Pik, where subscript i reflects the fact that patient i’s insurance coverage alters the retail price of drugs. Accordingly, physician j chooses drug k such that
Vijk - gi(Pik) ![]() Vijh - gi(Pih) for all k, hÎ
X.
Vijh - gi(Pih) for all k, hÎ
X.
Finally, remove the assumption that physicians are perfect agents of patients in terms of evaluating the cost of medications. I still assume that physicians genuinely care for the health status of their patients, therefore they do not intentionally undervalue the health benefits that their patients may obtain from the treatment. However, physicians may not be concerned with the cost of treatment that their patients incur. On the other hand, it is quite likely that physicians may be given an incentive to pay close attention to the cost of medications to insurance providers as well as to their patients due to the increasing effects of managed care policies. Define t j(× ,× ) as the function that measures the degree to which each physician is influenced by managed care measures and also internalizes the cost of drugs to her patients. The first argument in t j reflects the fact that managed care measures such as formularies are influenced by prices of drugs and the second argument in t j is the cost to patients. Thus, t j(× ,× ) is physician j’s evaluation on prices of drugs. Then, physician j chooses drug k such that
Vijk - t
j(Pik, gi(Pik)) ![]() Vijh - t
j(Pih, gi(Pih)) for all k, hÎ
X. (1)
Vijh - t
j(Pih, gi(Pih)) for all k, hÎ
X. (1)
The drug choice model described above naturally leads to an implementation of discrete choice models. First, the appropriateness of nested logit models to characterize demand for prescription drugs is discussed. Then, the advantages of mixed logit (random parameters logit) models over the nested logit models are discussed.
Nested Logit Model
One can argue that nested logit models are more appropriate than multinomial logit models for modeling the choice of prescription drugs by physicians (and patients, to some extent). Unobservable components of utilities of various drugs are likely to be correlated, since a brand name drug and its generic equivalents are likely to share unobserved attributes such as the molecule specific effects on individuals. Therefore, the independence from irrelevant alternatives property (IIA) is unlikely to hold.
One way to nest prescription choice given therapeutic class X is to have different molecules at the upper level and to have the choice of brand name drugs vs. generics at the lower level. This specification allows the utilities of drugs sharing the same molecule to be correlated so that various individual patient specific reactions to each molecule are accommodated. The structure in Figure 3 does not include the choice of generic products, though multiple generic products of the same molecule are often available. Since data used in this study provide prescription records where a physician notes either a brand name or a generic name, the data are not sufficient to determine exactly which generic product was dispensed to a patient if the record indicates the generic name. The pharmacist of the patient may ultimately choose one product if there exist several equivalent generic products. Therefore, the nesting structure in Figure 3 is two-level instead of three-level (in which the lowest cluster could be a choice of products). Also, some molecules are not available in generics due to patent protection (e.g., m3), while others may have multiple brand name drugs due to licensing agreements and/or the collaboration of several firms in research and development (e.g., m2).
Then, the nested logit model estimates the probability of drug k of molecule y being chosen in the therapeutic class X as a product of the conditional probability that drug k is chosen given the choice of molecule y times the marginal probability that molecule y is chosen. That is,
P(k) = P(k |my) ×
P(my) = 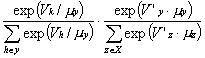 ,
,
where V. is the level of utility due to observable attributes for a corresponding drug, ![]() , and m
×
is an inclusive value coefficient. The inclusive value is consistent with the assumption of the nested logit when between zero and one, and the similarity of alternatives within the nest decreases as it approaches one (i.e., a low inclusive value implies that drugs of the same molecule are closer substitutes than drugs of various molecules).
, and m
×
is an inclusive value coefficient. The inclusive value is consistent with the assumption of the nested logit when between zero and one, and the similarity of alternatives within the nest decreases as it approaches one (i.e., a low inclusive value implies that drugs of the same molecule are closer substitutes than drugs of various molecules).
The other way to nest the prescription choice is to have the choice of brand name drugs vs. generics at the upper level and to have the choice of molecules at the lower level. This specification is based on the assumption that the utilities of brand name (generic) drugs are correlated. In other words, decision makers’ tastes for brand name (generic) drugs can be captured in this model. If heterogeneity among molecules is sufficiently small, this specification is the appropriate model to estimate demand for drugs.
One technical limitation of the nested logit model is that it does not allow correlations of random utility components (error terms) over molecules and the type of drugs (branded or generic drugs) simultaneously. In the specification in which the top level nest is the choice of molecule, the error term of utility is assumed to consist of an element attributable to the molecule (e m) and a random element (e mb); i.e., Umb = Vmb + e m + e mb. On the other hand, in the specification in which the top level nest is the choice between branded and generic drugs, the error term of utility for a drug is assumed to consist of an element attributable to the type of products (e b) and a random element; i.e., Umb = Vmb + e b + e mb. Therefore, if error terms of drugs sharing the same molecule are correlated as well as error terms of brand name drugs consisting of different molecules, i.e., Umb = Vmb + e m + e b + e mb, then the assumption of the nested logit model is violated and the estimates are biased and inconsistent. Mixed logit models allow more general structures of error terms; hence, they can accommodate the situation in which the assumption of the nested logit model is violated. Another advantage of the mixed logit model is that it can explicitly account for correlations of error terms arising from repeated observations of the same physician. The nested logit estimates are not asymptotically efficient, since information from the correlated error terms are not exploited. The issue on correlated error terms will be further discussed in the next section.
Mixed logit models assume that tastes of decision makers vary in the population. That is, coefficients of attributes of alternatives and characteristics of decision makers are not assumed to be the same but distributed across the population. Therefore, each decision maker’s coefficients, b
j, differ from the population mean, b , by some unobserved amount. This difference adds more randomness to the regular logit models and the multinomial logit equation is modified toPj(k) = 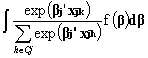 (2)
(2)
where j stands for a decision maker, k for a choice of drug, Cj for a choice set of decision maker j, x is a vector of explanatory variables, b j is a vector of coefficients for decision maker j and f(× ) is a density function of coefficients. This model is appealing as it accounts for correlated error terms that arise from unobservable shared attributes of drugs (i.e. preferences for generics). Also, the mixed logit model accommodates correlated error terms that arise from repeated observations of the same physician by varying coefficients of some variables over physicians. However, the estimation of this model is complex since no closed form solution exists. The model of equation (2) can be estimated following the procedure suggested by Revelt and Train (1996).
Suppose that physician j chooses a drug in each of T time periods. The utility from choice of drug k is Ujkt= b j’xjkt + e jkt, where xjkt is a vector of explanatory variables and e jkt is an unobservable term. The b j is a coefficient vector for physician j and is unobserved. If one knows the true value of b j, then the probability that physician j’s observed sequence of choices (conditional on b j) is
CPj(b
j) = 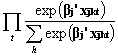 .
.
The value of b j is not known, but varies across population with density f(b |q *) where q * denotes true parameters (means and variances of b s). Then, the marginal probability is the integral of the above equation over all possible values of b s.
MPj(q
*) = ![]() .
.
Since q * is unobservable, q * needs to be estimated. This can be achieved by maximizing a simulated log likelihood function. For instance, a value of b is drawn from its distribution, given q , and CPj is calculated from this b . This process is repeated for many draws and the average of CPj over the repeated draws is the approximated MPj. That is,
SMPj(q
) = ![]() ,
,
where D is the number of draws and b d|q is the d-th draw from the distribution f(b |q ). The SMPj(q ) is an unbiased estimator of the UPj(q ). Then, the q * can be estimated as the q that maximizes the simulated log likelihood function,
SLL(q
) = ![]() .
.
Note that the mixed logit model can be viewed as Ujkt= b ’xjkt + g j’xjkt + e jkt, where the coefficient vectors are broken down to its mean b and deviations g j. Compared to the multinomial logit model, the mixed logit allows the unobservable portion of the utility (g j’xjkt + e jkt) to be correlated over drugs and repeated choices of a decision maker. For instance, the nested logit model can be replicated by entering molecule dummy variables (or a generic dummy variable) and allowing the coefficient of these dummy variables to vary over decision makers. The variances of these dummy variables are equivalent to the correlated unobserved portion of utility across all drugs within the molecule group (generic group) assumed in the nested logit model. By entering molecule dummy and generic dummy variables simultaneously, the mixed logit can also combine the two nested structures described in the previous sections.
If error terms are correlated in several different directions (e.g., correlation over un-nested sets of drugs), the mixed logit estimates correct biased nested logit estimates. Even if the correlation over drugs does not exist, the mixed logit estimates are more efficient than the nested logit estimates that do not utilize the information from correlated error terms of the same physician.
The limitations of the NAMCS data do not allow estimation using prices that each patient faces. Also, price coefficients are estimated for each insurance category in order to compare the differences in price elasticities. Therefore, equation (1) is modified to
Vijk - t
n(Pk, gn(Pk)) ![]() Vijh - t
n(Ph, gn(Ph)) for all k, hÎ
X. (3)
Vijh - t
n(Ph, gn(Ph)) for all k, hÎ
X. (3)
where index n denotes patient i’s insurance type, P. is the average retail price of a drug (note that subscript i is dropped from P), and t n(× ,× ) represents the degree to which physicians internalize the cost of medication.
A unit of observation is a prescription decision by a patient-physician combination per office visit. The variables used to represent Vijk in equation (3) are the age, sex, and race of the patient, marketing costs and age of the drug, and physician’s specialty. Since patient and physician characteristics do not vary across drugs, these variables need to be interacted with drug-specific dummy variables, molecule-specific dummy variables, or generic dummy variables to be included in the model. For example, a certain molecule may be more beneficial to a certain group of patients. Also, specialists may be more likely to place a high value on brand name drugs than primary care doctors do. In sum, equation (3) is estimated as:
Probij (k|X) = Probij (
Mka + YijIkb - Pkd n +e ijk= max Mha + YijIhb - Phd n +e ijh),where M. is a vector of drug characteristics including marketing cost and the age of medication as well as dummy variables for generics and subclasses of drugs; Yij is a vector of patient and physician characteristics including the age of patient i, dummy variables for the sex and race of patient i, and a dummy variable for a specialist; I. is a matrix of indicator variables which allows patient and physician characteristics to be interacted with a generic dummy variable and/or drug category dummy variables; P. is an average-price of medication; a and b are coefficient vectors, d n is a price coefficient; and e ij. is an unobservable element of utility. Table 1 lists definitions of the explanatory variables in the analysis.
Table 2 shows the coefficient estimates of the multinomial logit model where the unobserved portion of utility is assumed to be independently and identically Gumbel distributed. There is no grouping of drugs in this model. The price coefficients are estimated separately for four insurance groups. The price coefficients are affected by various factors. If one assumes that a physician takes into consideration the drug expenses of her patient, then the existence of a co-payment reduces the magnitude of the price coefficients for the insured sector. In particular, Medicaid patients are generally fully covered for their prescription consumption. Therefore, the price coefficient for Medicaid is expected to be zero if physicians care only for what patients pay and there were no managed care measures. On the other hand, increased sensitivity towards drug prices due to managed care cost-cutting measures should put upward pressure on the magnitude of price coefficients for the HMO and Medicaid sectors and to some extent in the commercial sector (as the commercial sector includes patients with managed care benefits). Therefore, without agency problems and managed care measures, one can expect the price coefficient for uninsured patients to be the most negative. The existence of managed care measures should reduce the differences of price coefficients between Medicaid/HMO and self-paid sectors.
The results of the basic specification are shown in column (1). All price coefficients are negative and the magnitude of price effects on the choice of drugs is the greatest for the Medicaid sector, followed by HMO, and commercial plans. The least negative price coefficient for the self-paid sector suggests the existence of an agency problem. However, one should be aware of other factors which may affect the price sensitivity in interpreting these results. For instance, one may argue that uninsured patients have more severe conditions than insured patients, therefore they need more expensive medications. Given the fact that the efficacy rates of all antidepressant drugs are similar and differences arise from the extent of side effects, there is no compelling reason to believe the uninsured need more expensive medications than others. Another factor affecting the price coefficient result might be that average income of self-paid patients is higher than that of insured patients. The inclusion of income variables could help explain the parameter estimates, but income information is unfortunately not available in the NAMCS data. Although there are no apparent reasons to believe that uninsured patients are wealthier than insured patients, one should be aware of the possible income effect while interpreting the result as the existence of agency problems.
The results in price coefficients also confirm the hypothesis that managed care measures implemented by the Medicaid and HMO plans are effective. Since the substitution of generics for brand name drugs with expired patents at the dispensing stage is particularly common for the Medicaid and HMO sectors, one can expect doctors writing prescriptions for patients in those sectors to have less incentive to note the exact generic names on prescription pads. Therefore, the generic substitution policy may reduce doctors’ sensitivity to prices of certain brand name products, possibly attenuating the price coefficients. The greater magnitude of price coefficients in the managed care sectors, despite the possible effect of generic substitution rules, suggests that cost-cutting measures have significant influence on physician behavior as well as on pharmacist behavior.
The t-statistics for testing the hypothesis that any of the price coefficients differ from each other are computed. It is found that the price coefficient of the Medicaid sector is significantly different from that of any other sector, though the differences between the HMO, commercial plans, and self-paid sectors are not statistically significant at a ten percent level. A possible explanation for the lack of significant differences in the price coefficients of the HMO and commercial sectors is that the commercial plans include patients with managed-prescription coverage such as card programs. As pointed out in Section 4, due to the limitation of the NAMCS data, the commercial plan category is not equivalent to the un-managed drug benefits category. This fact might have reduced the gaps between the HMO sector and the commercial sector. Nonetheless, the commercial category seems less price sensitive than the HMO sector.
The sign of the coefficients for the log of advertising costs is positive, as expected. The advertising cost variable was originally included in the model, but taking a log of the advertising costs improved the fit of the model dramatically, indicating decreasing marginal returns to advertising. Also, the positive coefficient on the log of the age of product implies that physicians are more comfortable in prescribing drugs with a long history but the marginal effect dissipates as products get older. This is a reasonable result considering that the longer a drug has been on the market, the more information on its efficacy and side effects is available. Although one can argue that older drugs are less advanced than newer drugs, drug category variables are expected to capture the quality differences of different classes of drugs.
It is also shown in Table 2 that non-whites are more likely to be prescribed generics and females are more likely to be prescribed brand name drugs. A possible explanation for the fact that women are more likely to be prescribed branded products may be related to the fact that women are more sensitive to the side effect of weight gain. As presented in Table A.1 of the Data Appendix, many molecules which are available in both branded and generic drugs have a higher risk of weight gain. In turn, women may have a tendency to choose newer drugs with reduced risk of weight gain, which are not available in generic versions. The negative coefficient on the specialist dummy variable indicates that specialists tend to prescribe brand name drugs. This is consistent with the notion that specialists are more familiar with scientific research supporting the efficacy and safety of drugs and that the research often focuses more on brand name drugs due to pharmaceutical company sponsorship.
Four constants are included in the model instead of twenty-seven product-specific constants. Since price, marketing costs, and the age of product are product specific, the inclusion of product-specific constants causes near-multicollinearity. The category constants attempt to capture any quality differences among various types of products. As pointed out in Section 4, there are three categories in the antidepressant market: Tricyclics and Heterocyclics, SSRIs, and Others. The Tricyclics and Heterocyclics are the base category in the estimation. The Anafranil constant is also added, since Anafranil is the only drug indicated for obsessive compulsive disorder. The fewer adverse effects associated with the SSRIs are reflected on the positive SSRI coefficient. The positive generic coefficient indicates that people like generics more than brand name drugs, given that other things are equal. This is contrary to the typical view that the quality of brand name drugs is superior to generics in terms of inert fillers, preservatives, coloring agents, binders, etc. It is possible that managed care policies encouraging the utilization of generics may be reflected in the positive generic coefficient.
The specification in column (2) is an attempt to investigate the effects of generic substitution at the dispensing level on physician prescription behavior. Since the NAMCS data are prescription data, the information on what was ultimately consumed is not obtainable. Some prescriptions where brand name products were chosen in the NAMCS data surely resulted in the substitution for generics due to generic substitution policies (e.g., this can be interpreted as a classification problem). When a physician writes a prescription for a brand name drug no longer under patent protection, her expectation on the price to be paid is not necessarily the price of the brand name drug. The physician may assume the price to be the price of the generic equivalent or the combination of two (i.e., the weighted average where the weights are probability of each drug to be chosen). In a sense, one may think that the price of brand name drugs which are available in generics is "overstated" in the model, since brand names are always more costly than generics. Without information on the substitution rates for each insurance category, the price of non-patented brand name products cannot be appropriately modified to the weighted average of the prices of the brand name and its generic equivalent.
One way to deal with this problem given the restriction of data is to specifically allow the price of a non-patented brand name drug to have less impact on utilities than those of patented and generic drugs. For instance, the effect of a one dollar difference between two patented products is more significant than a one dollar difference between a non-patent protected brand name and its generic equivalent in prescription decisions due to the possibility of generic substitution. In other words, one can expect the price change of non-patent-protected brand name products to have smaller influence on physician prescription behavior than the price change of other products. In order to account for this effect in the model, the dummy variable for non-patented brand name products is interacted with prices. Since this price variable is specific to non-patented brand name drugs, it should function as an adjustment factor for the price coefficient of non-patented brand name drugs. The coefficients of the price adjustment variables for non-patented brand name drugs are expected to be positive and smaller in magnitude than the price coefficients, because the price effect of non-patented brand name drugs is believed to be smaller than the price effect of other products.
Column (2) shows the results of this specification. The coefficients of price adjustment variables are all positive and smaller in magnitude than the price coefficients. Also, all price coefficients became more negative in agreement with the predictions above. The results confirm the effect of generic substitutions on physician prescription behavior. One interesting result is that the price adjustment coefficient for HMOs is not statistically significant. Since many HMOs provide formulary compliance incentives to physicians, the results reflect the possibility that physicians at HMOs are more likely to precisely note generic names on their prescription pads when they do not find a specific reason to prohibit the use of generics. On the other hand, physicians seeing Medicaid patients seem to delegate the choice of branded or generic versions of the same molecule to pharmacists.
The parameter estimates of the nested logit models are shown in Table 3. Model A describes the model where the upper level is the choice of molecules and the lower level is the choice of drugs (e.g., pioneer brand = the first producer of the molecule, the 2nd brand = the branded drug that is the 2nd entrant, and generics). Model B describes the model where the nesting structure is reversed. Only the results from the second specification with price adjustment variables are listed. Most coefficients in both models are qualitatively similar to those in Table 2, except for the coefficients of the price adjustment variables for all four insurance categories. Here, these coefficients become insignificant. In model A, the scale coefficient is above one, which is not in agreement with the requirement of the nested logit model that it be between zero and one. The scale coefficient in model B is 1.094, which is also greater than one although it is very close to one. As explained in Section 5, model A allows the utilities of drugs of the same molecule to be correlated, whereas model B allows the utilities of brand name (generic) products to be correlated. The weakness of nested logit models is that they only allow one of these possible correlations in the same model and that multi-dimensional correlations are not captured. The size of scale coefficients implies that the assumptions of both models are violated. It appears that the correlation structures of error terms are multi-dimensional and both types of correlations need to be included. Mixed logit models eliminate the restrictive assumption on the structure of error terms placed by the nested logit model and explicitly account for both types of correlations: the correlations over molecules and the type of products. This can be done by allowing coefficients of molecules and generic dummy variables to vary over the sample data. Further, allowing coefficients to vary across physicians captures correlated error terms for multiple observations of the same physician in the sample data.
Table 4 shows the result of mixed logit estimation using 500 repetitions. In order to replicate the assumption of model A and model B simultaneously, the coefficients of molecule- specific dummy variables (for the molecules that are supplied by multiple drugs) and the generic dummy variable should be estimated to be random. However, the use of molecule dummy variables in the model, with the generic constant, mimics the effects of drug-specific dummy variables. Since prices, advertising costs and the age of products are drug-specific variables, the addition of molecule dummy variables creates near-multicollinearity. One solution to this problem is to allow the coefficients of three drug category dummy variables, included to capture the quality effects, to vary over the sample. This specification allows the situation such as the case where a decision maker who likes Prozac also likes Zoloft. This is shown in model C. The other method to avoid the problem of near-multicollinearity is to constrain the mean of the molecule-specific dummy variables to be zero and estimate the standard deviation of those dummy variables. This specification allows an individual’s taste for a certain molecule to vary, while it assumes that the taste variations cancel out each other on average. In order to reduce the number of parameters, the standard deviation of the molecule specific constants are constrained to equal each other. The results are shown in model D.
The results of models C and D are fairly similar except for the coefficients of price adjustment terms. The coefficients of price adjustment terms are not significant and the HMO sector has a negative coefficient. This is probably due to the fact that the addition of molecule specific constants are somewhat collinear with price adjustment terms since they both enter in the equation only if the molecule is available in multiple drugs. Nonetheless, the adjusted likelihood ratio index is higher for model D and the likelihood ratio test rejects the hypothesis that model C and model D are equivalent.
The coefficient estimates of the mixed logit model are qualitatively comparable to the results obtained in the multinomial logit models, although the Hausman/McFadden tests reject the IIA assumption and the standard deviations of random coefficients are large relative to the standard errors. The price coefficients are negative and the Medicaid sector still exhibits the greatest price effect. The differences between the Medicaid and commercial sectors and between the Medicaid and self-paid sectors are significant, whereas the differences among the rest of the combinations are not statistically significant. The main change from the previous models is that the specialist coefficient is no longer significant. Instead, the age coefficient is positive and significant, indicating the tendency for older patients to be prescribed generics.
The mean of the SSRI constant is positive and the standard deviation of 1.091 and 0.763 in models C and D, respectively, implies that almost one hundred percent of the sample population place a positive value on the SSRIs. The mean and standard deviation of the Anafranil and other constants exhibit greater taste variations in the sample data, as thirty-six percent and forty percent of the sample population have negative coefficients, respectively. This result is reasonable in that the usefulness of these drugs depends on each patient’s specific conditions. The generic coefficient is positive and significant as in the multinomial and nested logit models. However, the mixed logit model reveals that twenty-two percent of the sample population place negative generic coefficients in model C and thirty-three percent in model D. The largest estimated standard deviation of the generic constant implies that preferences for generics vary among the population more than preferences for a certain molecule and/or category group.
Simulated Price Elasticities
The own- and cross-price elasticities (i.e., the percentage change in the share of a product chosen due to a one percent change in its price or its competitor’s price) are calculated from estimates of the mixed logit model D. For each insurance category, the share of each product is computed after a ten percent change in the price of each product, and the elasticities are computed from the change in shares. Since the mixed logit allows taste variations over decision makers, the probability of each product chosen is approximated by taking an average of 2000 draws for normally distributed coefficients. Table 5 shows the elasticity of a product in the jth column with respect to the price change of a product in the ith row. If there is only one branded drug for a particular molecule, it is classified as "monopoly – pioneer brand." If the molecule is available in multiple drugs, then the branded drug that was produced first is referred as "non-monopoly – pioneer brand," the 2nd branded drug as "non-monopoly – 2nd brand," and generics as "non-monopoly - generics." These price elasticities are the weighted average of an elasticity of each product, where the weights are the share of the product before the price change.
The Medicaid sector exhibits the highest own-price elasticities of demand in general. Also, brand name products, particularly monopoly brand names, are considerably more price elastic than generics across all insurance categories. The weighted average own-price elasticities for generics range from -0.35 to -0.40, whereas those for brand names range from -1.06 to -1.91. If one sees the demand curve for antidepressant drugs as a summation of each consumer, then brand name products are purchased by consumers with a high willingness to pay, and generics are purchased by the patients with a low willingness to pay. Therefore, brand name products capture the elastic portion of the demand curve and generics the inelastic portion of the demand curve. In particular, the manufacturers of patented brand name products are monopolists and face a downward sloping demand curve for their products. As profit maximization suggests, they seem to choose prices which correspond to the elastic portion of the demand curve.
The results for cross-price elasticities reveal some substitution patterns of different products. When there are multiple drugs of the same molecule, the cross-price elasticities between branded and generic drugs of the same molecule are greater than those between therapeutic substitutes. For instance, if the price of a generic drug changes, the shares of brand name drugs of the same molecule change more than the share of another drug of the different molecule on average. Also, the price increase of a brand name drug raises the share of its generic equivalent more than that of other drugs of a different molecule. This implies higher substitutability between drugs of the same molecule than across drugs of different molecules, which is consistent with the findings of Ellison et al. (1997). When there is only one product for a particular molecule, the cross-price elasticities can be calculated only between products of different molecules. The result shows that the substitutability among monopoly branded drugs is the highest of all, indicating that the newer brand name drugs whose patents have not expired compete with each other. One explanation for this result is that patients who can afford and are taking a relatively expensive monopoly brand name drug consider another monopoly brand name drug to be a closer substitute than other older brand name drugs (e.g., Zoloft may be considered as a closer substitute of Prozac than Elavil).
Only the results from the mixed logit models are presented in Table 6. The inclusion of price adjustment variables improves the likelihood ratio index and t-statistics for the price coefficients more substantially for beta blockers than for antidepressants. The price adjustment coefficients are all positive and significant except for the HMO coefficient in model D, implying that the price effect of non-patented products is smaller than other products. In particular, the Medicaid sector has the largest coefficient. The Medicaid sector also has the largest price adjustment coefficient in the antidepressant market. This result is consistent with the fact that many Medicaid programs implement a generic substitution policy in which pharmacists are given financial incentives to dispense generics. Physicians know that their brand name prescriptions are very likely to be substituted for generics, thus, they may not precisely note generic names on prescriptions. The size of the price adjustment coefficient estimate for the Medicaid sector is consistent with the view that this physician behavior, based on the anticipation of generic substitutions, is particularly true for the Medicaid sector. On the other hand, the insignificant coefficient of the price adjustment term for the HMO sector indicates that physicians are more likely to note a generic name rather than a brand name on prescription pads despite the generic substitution policy at the dispensing level often enforced by HMOs. This finding is consistent with the study of Hellerstein (1994).
For patented brand name and generic products, the Medicaid price coefficient is the most negative, followed by the HMO, commercial, and self-paid sectors. However, they are not significant for the self-paid sector in either model and for the commercial sector in model C. The differences of the price coefficients between the HMO/Medicaid sectors and the commercial/self-paid sectors are statistically significant except for the difference between the Medicaid and commercial sector, which has a t-statistic of 1.58. Therefore, the effect of prices in the HMO and Medicaid sectors is significantly greater than the effect in the self-paid sector and is generally greater than the effect in the commercial sector. This result is consistent with the findings from the antidepressant market, where the price coefficient of the Medicaid sector significantly differs from those of the commercial and self-paid sectors.
In addition to the four variables constructed from patient and physician characteristics and used for the antidepressant market, two more patient-specific variables are added: obesity and smoking dummy variables. Since obesity and smoking are both crucial risk-increasing factors for hypertensive patients, these variables are interacted with the multi-indication dummy variable (angina constant). Although obesity and smoking status do not necessarily cause angina, these conditions can increase the chance of disease complication. Some physicians are often cautious choosing a drug for multiple purposes rather than a single purpose if they see a chance of complication which may not be apparent today. The coefficients on these patient and physician specific variables are not significant, though the smoking coefficient is positive and almost significant at a ten percent confidence level.
The advertising costs and age of a product have strong effects on the choice of drugs in the beta blocker market as in the antidepressant market.
The generic and angina constants are both positive, yet there is a fair amount of taste variation. Twenty-two percent of the sample population in model C and seventeen percent in model D dislike generics. This taste variation in generic products is similar to the result from the antidepressant market. Twenty-two percent of the sample population also place a negative coefficient on drugs with multiple indications. In addition, the estimate of standard deviation of molecule specific constants is significant, confirming that the benefits from products of the same molecule are correlated.
Simulated Price Elasticities
Table 8 provides own- and cross-price elasticities for beta blocker drugs calculated from the estimates of the mixed logit model D. In Table 8, own-price elasticities are all negative except for non-monopoly pioneer drugs in the self-paid sector. This is due to the fact that the price coefficient for non-monopoly brand name products is positive (-0.065+0.714=0.649). In all sectors, monopoly brand name drugs are most price elastic as in the case for the antidepressant market. Also, the HMO and Medicaid sectors exhibit higher elasticities than the commercial and self-paid sectors. One notable difference from the antidepressant market is that the second branded drug is more elastic than non-monopoly pioneer drugs. Since there is only one second branded drug in the beta blocker market and its molecule is not available in generic format, one should note that the second branded drug should be viewed similar to monopoly pioneer drugs. Contrary to the antidepressant market, non-monopoly brand name drugs are less price elastic than generics in the Medicaid, commercial, and self-paid sectors. This result suggests that the effect of generic substitutions at the dispensing level on physicians’ prescription behavior is significant in the beta blocker market. That is, generic substitutions are probably more common in the beta blocker market than in the antidepressant market.
The estimates of cross-price elasticities here are somewhat different from the findings in the antidepressant market. When there are multiple products sharing the same molecule, the price increase of a brand name product leads to a greater change in the share of other products of the same molecule than that of a different molecule. However, the price change of a generic drug affects the share of another generic drug of a different molecule more than that of the branded drug sharing the same molecule. Therefore, a generic drug loses its market share to other generics relatively more than its branded equivalent. When there is only one branded drug for a molecule, then the effect of price change of the monopoly branded drug is greater on the share of other monopoly branded drugs than those of non-monopoly branded drugs and generics. This result is consistent with findings in the antidepressant market.
This paper examines the effects of insurance coverage on demand for prescription drugs in the antidepressant and beta blocker markets. The analysis of the micro-level data on a national sample of patient office visits reveals that uninsured patients are the least sensitive to prices of drugs. Given the fact that they have to pay for the entire cost of drugs, this is a surprising result. It suggests that a physician may not be a good agent for her patient, in terms of evaluating the effect of drug expenses on her patient. Another finding is that the substitutability between drugs based on the same molecule is higher than the substitutability across different molecules, except for beta blocker generics, for which generics of different molecules seem to be closer substitutes than their brand name equivalents. Therefore, drugs sharing the same molecule are in general closer substitutes than therapeutic substitutes. In addition, relatively newer drugs, which are the only product of a certain molecule, are found to compete against each other more than against older, non-patented drugs. This therapeutic substitutability between newer drugs seems to be higher than the substitutability between drugs sharing the same molecule.
Another important result shows the effectiveness of managed care measures in increasing the sensitivity of physicians towards drug costs. Demand in the Medicaid sector is found to be the most price elastic in the antidepressant market, and both the Medicaid and HMO sectors have more elastic demand than the commercial and self-paid sectors in the beta blocker market. Also, generic substitution policies seem to affect physicians’ prescription behavior. Physicians delegate some responsibilities to pharmacists in choosing branded or generic versions of the same molecule, but it appears to be much less pronounced in the HMO sector.
In this study, information on the outcomes of drug therapies is not available. Therefore, it is not feasible to determine whether or not managed care sectors achieve satisfactory health outcomes more economically than other sectors. It is quite possible that the health outcomes of patients might decline with the aggressive utilization of cost-cutting measures. For instance, tight restrictions on the use of certain medications may lead physicians to choose drugs that are not optimal treatments for some patients. Another possible effect of managed care measures, which is beyond the scope of this study, is the increased rate of substitution between drug and non-drug therapies. Due to restrictions on drug use, physicians may substitute expensive non-drug treatments for drug therapies, even though non-drug treatments may not provide any advantages in terms of efficacy. Thus, aggressive cost-cutting incentives in prescribing drugs may lead to increase in overall medical care costs. This situation is most likely to occur for patients with an indemnity health plan plus managed prescription drug coverage. It would be valuable to analyze differences in the health outcomes between the managed and non-managed care sectors and explore the degree of substitutability between drug and non-drug therapies as a function of the insurance type.
In today’s environment where managed care sectors continue to grow, the results of my study suggest that pharmaceutical manufacturers will increasingly compete on price, rather than relying on their traditional marketing strategies of mobilizing large sales forces. Consequently, manufacturers’ profit opportunities in the future may not be as generous as in the 1980s and the early 1990s. The slowdown of price increases in prescription drugs may have two effects on R&D spending by pharmaceutical manufacturers. First, R&D spending could decline, resulting in fewer discoveries of new drugs. That is, the rise of managed care pharmacy may reduce the cost of medication today, but it may do so by sacrificing the benefits from potential future products. Second, the downward pressure on prices could change the type of research projects pursued by manufacturers. For instance, manufacturers may be more likely to invest in research directed at developing breakthrough drugs that establish a new therapeutic class, rather than "me-too" branded drugs that enter an established therapeutic class. Since the use of formularies and other cost-cutting measures increases price elasticities and the competition among drugs of similar efficacy, the attractiveness of participating in the markets with incumbents is lowered. The relative size of the payoff from being the first branded entrant to that of the second or third branded entrant in a therapeutic class becomes greater as the managed care sector grows. There should be more emphasis on the research for breakthrough drugs than "me-too" drugs which do not offer many therapeutic advantages over the incumbent. It would be interesting to evaluate how the growth of managed care pharmacy may not only alter the size of pharmaceutical R&D spending but also change the way funds for R&D are allocated.
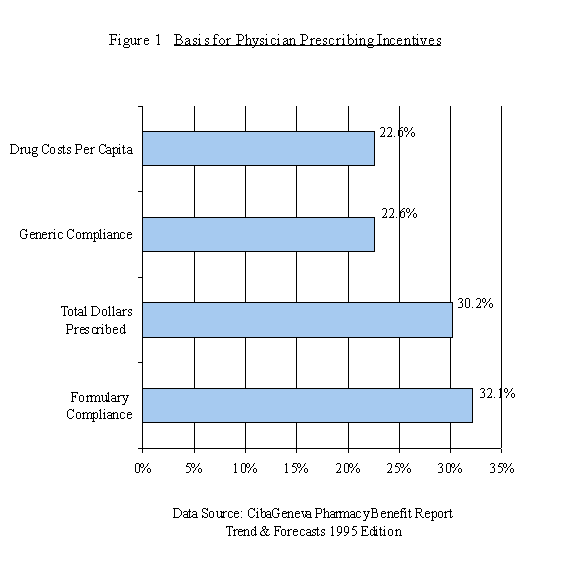
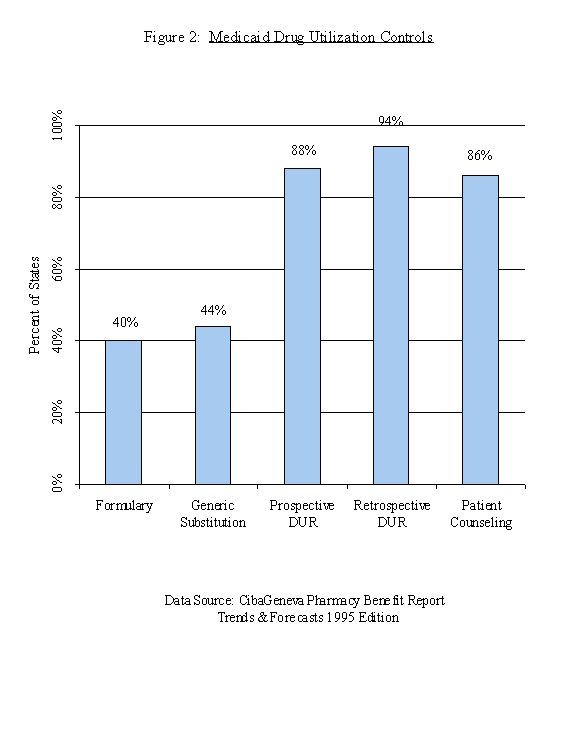
Figure 3: Example of the Nesting Structure
Table 1: Variable Definitions
|
Variable names: |
Definitions: |
|
Price for HMO |
Average retail price of a drug per day (CPI adjusted) interacted with dummy for HMO patients. |
|
Price for Medicaid |
Average retail price of a drug per day (CPI adjusted) interacted with dummy for Medicaid patients. |
|
Price for Comm. Plan |
Average retail price of a drug per day (CPI adjusted) interacted with dummy for patients covered by a commercial plan. |
|
Price for Self-paid |
Average retail price of a drug per day (CPI adjusted) interacted with dummy for uninsured patients. |
|
Price adjustment for HMO |
Average retail price of a drug per day (CPI adjusted) interacted with dummy for branded drugs for which the generic equivalents are available and dummy for HMO patients. |
|
Price adjustment for Medicaid |
Average retail price of a drug per day (CPI adjusted) interacted with dummy for branded drugs for which the generic equivalents are available and dummy for Medicaid patients. |
|
Price adjustment for Comm. plan |
Average retail price of a drug per day (CPI adjusted) interacted with dummy for branded drugs for which the generic equivalents are available and dummy for patients covered by a commercial plan. |
|
Price adjustment for Self-paid |
Average retail price of a drug per day (CPI adjusted) interacted with dummy for branded drugs for which the generic equivalents are available and dummy for uninsured patients. |
|
Log(Advertising cost) |
Natural log of advertising cost of a drug in thousands of dollars (pharmaceutical PPI adjusted). |
|
Log(Age of product) |
Natural log of the number of years for which a drug has been available. |
|
Age*Generics |
Age of the patient interacted with dummy for generics. |
|
Non-white*Generics |
Dummy for non-white patients interacted with dummy for generics. |
|
Female*Generics |
Dummy for female patients interacted with dummy for generics. |
|
Specialist*Generics |
Dummy for specialists interacted with dummy for generics. |
|
Generic constant |
Dummy for generics. |
|
Anafranil constant |
Dummy for Anafranil (antidepressant drug) |
|
Other constant |
Dummy for antidepressant drugs in the "other" category. |
|
SSRI constant |
Dummy for antidepressant drugs in the "SSRI" category |
|
Obesity*Angina |
Dummy for obese patients interacted with dummy for beta blocker drugs whose indication includes angina. |
|
Smoking*Angina |
Dummy for smoking patients interacted with dummy for beta blocker drugs whose indication includes angina. |
|
Angina constant |
Dummy for beta blocker drugs whose indication includes angina. |
|
Molecule-specific constants |
Dummies for molecules which are available in multiple drugs. |
Table 2: Multinomial Logit Results for Antidepressants
|
(1) |
(2) |
||||
|
Variable name |
Estimate |
Standard error |
Estimate |
Standard error |
|
|
1 |
Price for HMO |
**-0.780 |
0.086 |
**-0.910 |
0.096 |
|
2 |
Price for Medicaid |
**-0.927 |
0.093 |
**-1.064 |
0.103 |
|
3 |
Price for Comm. Plan |
**-0.744 |
0.071 |
**-0.876 |
0.084 |
|
4 |
Price for Self-paid |
**-0.668 |
0.083 |
**-0.801 |
0.094 |
|
5 |
log(Advertising cost) |
**0.351 |
0.014 |
**0.361 |
0.015 |
|
6 |
log(Age of product) |
**1.387 |
0.048 |
**1.366 |
0.048 |
|
7 |
Age * Generics |
0.003 |
0.002 |
0.003 |
0.002 |
|
8 |
Non-white * Generics |
0.275 |
0.156 |
0.281 |
0.157 |
|
9 |
Female * Generics |
*-0.215 |
0.086 |
*-0.214 |
0.086 |
|
10 |
Specialist * Generics |
**-0.219 |
0.085 |
**-0.219 |
0.085 |
|
11 |
Generic constant |
**1.841 |
0.163 |
**1.925 |
0.166 |
|
12 |
Anafranil constant |
**1.633 |
0.152 |
**1.882 |
0.171 |
|
13 |
Other constant |
**0.623 |
0.065 |
**0.664 |
0.065 |
|
14 |
SSRI constant |
**2.812 |
0.083 |
**3.044 |
0.106 |
|
15 |
Price adjustment for HMO |
0.085 |
0.074 |
||
|
16 |
Price adjustment for Medicaid |
**0.231 |
0.086 |
||
|
17 |
Price adjustment for Comm. Plan |
**0.190 |
0.052 |
||
|
18 |
Price adjustment for Self-paid |
*0.135 |
0.069 |
||
Number of observations 3397 3397
Log of likelihood function -9045.81 -9036.33
Adjusted likelihood ratio index 0.185 0.186
** 1% significant level
* 5% significant level
Table 3: 2-level Nested Logit Results for Antidepressants
Model A: Upper-level: the choice of molecules;
Lower-level: the choice between pioneer brand name, the second brand name,
and generic drugs.
Model B: Upper-level: the choice between pioneer brand name, the second brand name,
and generic drugs;
Lower-level: the choice of molecules.
|
(A) |
(B) |
||||
|
Variable name |
Estimate |
Standard error |
Estimate |
Standard error |
|
|
1 |
Price for HMO |
**-0.954 |
0.105 |
**-0.954 |
0.116 |
|
2 |
Price for Medicaid |
**-1.082 |
0.111 |
**-1.115 |
0.123 |
|
3 |
Price for Comm. Plan |
**-0.934 |
0.089 |
**-0.916 |
0.105 |
|
4 |
Price for Self-paid |
**-0.888 |
0.101 |
**-0.835 |
0.113 |
|
5 |
log(Advertising cost) |
**0.497 |
0.022 |
**0.423 |
0.030 |
|
6 |
log(Age of product) |
**1.642 |
0.060 |
**1.388 |
0.081 |
|
7 |
Age * Generics |
0.005 |
0.004 |
0.003 |
0.002 |
|
8 |
Non-white * Generics |
*0.502 |
0.239 |
0.278 |
0.157 |
|
9 |
Female * Generics |
*-0.279 |
0.132 |
*-0.215 |
0.086 |
|
10 |
Specialist * Generics |
**-0.797 |
0.133 |
**-0.225 |
0.085 |
|
11 |
Generic constant |
**2.506 |
0.251 |
**2.039 |
0.223 |
|
12 |
Anafranil constant |
**2.675 |
0.196 |
**1.495 |
0.186 |
|
13 |
Other constant |
**0.865 |
0.074 |
**0.518 |
0.083 |
|
14 |
SSRI constant |
**3.397 |
0.121 |
**2.687 |
0.124 |
|
15 |
Price adjustment for HMO |
0.006 |
0.099 |
-0.096 |
0.083 |
|
16 |
Price adjustment for Medicaid |
0.178 |
0.112 |
0.069 |
0.094 |
|
17 |
Price adjustment for Comm. Plan |
0.085 |
0.064 |
0.025 |
0.060 |
|
18 |
Price adjustment for Self-paid |
-0.002 |
0.089 |
-0.048 |
0.077 |
|
19 |
Scale coefficient |
**2.002 |
0.120 |
**1.094 |
0.043 |
Number of observations 3397 3397
Log of likelihood function -8963.7 -9050.21
Adjusted likelihood ratio index 0.192 0.184
** 1% significant level
* 5% significant level
Table 4: Mixed Logit Results for Antidepressants
Model C: Normally-distributed coefficients for Generic, Anafranil, Other and SSRI
constants.
Model D: Normally-distributed coefficients for Generic, Anafranil, Other and SSRI
constants & normally-distributed error components of molecule-specific
constants.
|
(C) |
(D) |
|||||||
|
Variable name |
Estimate |
Standard error |
Estimate |
Standard error |
||||
|
1 |
Price for HMO |
**-0.905 |
0.096 |
**-1.019 |
0.095 |
|||
|
2 |
Price for Medicaid |
**-1.072 |
0.106 |
**-1.184 |
0.120 |
|||
|
3 |
Price for Comm. Plan |
**-0.881 |
0.074 |
**-1.009 |
0.080 |
|||
|
4 |
Price for Self-paid |
**-0.875 |
0.090 |
**-0.970 |
0.094 |
|||
|
5 |
log(Advertising cost) |
**0.367 |
0.013 |
**0.429 |
0.017 |
|||
|
6 |
log(Age of product) |
**1.375 |
0.045 |
**1.605 |
0.041 |
|||
|
7 |
Age * Generics |
*0.006 |
0.003 |
*0.006 |
0.003 |
|||
|
8 |
Non-white * Generics |
*0.482 |
0.208 |
*0.507 |
0.236 |
|||
|
9 |
Female * Generics |
*-0.210 |
0.107 |
*-0.247 |
0.116 |
|||
|
10 |
Specialist * Generics |
0.132 |
0.173 |
0.180 |
0.156 |
|||
|
11 |
Generic constant |
Mean |
**1.187 |
0.224 |
**1.148 |
0.217 |
||
|
12 |
Standard deviation |
**1.527 |
0.114 |
**1.812 |
0.114 |
|||
|
13 |
Anafranil constant |
Mean |
0.641 |
0.431 |
**2.037 |
0.299 |
||
|
14 |
Standard deviation |
**1.769 |
0.290 |
**1.115 |
0.212 |
|||
|
15 |
Other constant |
Mean |
*0.271 |
0.116 |
**0.746 |
0.094 |
||
|
16 |
Standard deviation |
**1.077 |
0.116 |
**0.508 |
0.112 |
|||
|
17 |
SSRI constant |
Mean |
**2.848 |
0.133 |
**3.512 |
0.144 |
||
|
18 |
Standard deviation |
**1.091 |
0.088 |
**0.763 |
0.089 |
|||
|
19 |
Price adjustment for HMO |
0.054 |
0.078 |
-0.147 |
0.076 |
|||
|
20 |
Price adjustment for Medicaid |
**0.290 |
0.090 |
0.074 |
0.098 |
|||
|
21 |
Price adjustment for Comm. Plan |
**0.203 |
0.050 |
0.005 |
0.056 |
|||
|
22 |
Price adjustment for Self-paid |
*0.153 |
0.070 |
-0.039 |
0.080 |
|||
|
23 |
Molecule-specific constants |
Standard deviation |
**1.556 |
0.053 |
||||
Number of observations 3397 3397
Log of likelihood function -8758.32 -8411.09
Adjusted likelihood ratio index 0.214 0.241
** 1% significant level
* 5% significant level
Table 5: Simulated Price Elasticities for Antidepressants
|
HMO |
Own molecule |
Other molecules |
|||||||
|
Mono-poly |
Non-monopoly |
Mono-poly |
Non-monopoly |
||||||
|
Pioneer brand |
Pioneer brand |
2nd brand |
Generic |
Pioneer brand |
Pioneer brand |
2nd brand |
Generic |
||
|
Mono-poly |
Pioneer brand |
-1.585 |
0.185 |
0.131 |
0.126 |
0.086 |
|||
|
Non-mono-poly |
Pioneer brand |
-1.294 |
0.159 |
0.098 |
0.038 |
0.038 |
0.038 |
0.021 |
|
|
2nd brand |
0.123 |
-1.150
|
0.073 |
0.032 |
0.032 |
0.031 |
0.020 |
||
|
Generic |
0.032 |
0.032 |
-0.362 |
0.008 |
0.007 |
0.008 |
0.018 |
||
|
MEDICAID |
Own molecule |
Other molecules |
|||||||
|
Mono-poly |
Non-monopoly |
Mono-poly |
Non-monopoly |
||||||
|
Pioneer brand |
Pioneer brand |
2nd brand |
Generic |
Pioneer brand |
Pioneer brand |
2nd brand |
Generic |
||
|
Mono-poly |
Pioneer brand |
-1.909 |
0.185 |
0.124 |
0.122 |
0.081 |
|||
|
Non-mono-poly |
Pioneer brand |
-1.226 |
0.172 |
0.109 |
0.045 |
0.043 |
0.042 |
0.024 |
|
|
2nd brand |
0.132 |
-1.184
|
0.081 |
0.036 |
0.035 |
0.034 |
0.022 |
||
|
Generic |
0.036 |
0.037 |
-0.402 |
0.010 |
0.008 |
0.009 |
0.021 |
||
Table 5: Simulated Price Elasticities for Antidepressants (Continued)
|
COMMERCIAL |
Own molecule |
Other molecules |
|||||||
|
Mono-poly |
Non-monopoly |
Mono-poly |
Non-monopoly |
||||||
|
Pioneer brand |
Pioneer brand |
2nd brand |
Generic |
Pioneer brand |
Pioneer brand |
2nd brand |
Generic |
||
|
Mono-poly |
Pioneer brand |
-1.596 |
0.175 |
0.121 |
0.119 |
0.081 |
|||
|
Non-mono-poly |
Pioneer brand |
-1.136 |
0.152 |
0.097 |
0.038 |
0.038 |
0.037 |
0.021 |
|
|
2nd brand |
0.115 |
-1.073
|
0.071 |
0.031 |
0.031 |
0.30 |
0.019 |
||
|
Generic |
0.029 |
0.030 |
-0.356 |
0.008 |
0.007 |
0.007 |
0.017 |
||
|
SELF-PAID |
Own molecule |
Other molecules |
|||||||
|
Mono-poly |
Non-monopoly |
Mono-poly |
Non-monopoly |
||||||
|
Pioneer brand |
Pioneer brand |
2nd brand |
Generic |
Pioneer brand |
Pioneer brand |
2nd brand |
Generic |
||
|
Mono-poly |
Pioneer brand |
-1.522 |
0.174 |
0.122 |
0.119 |
0.082 |
|||
|
Non-mono-poly |
Pioneer brand |
-1.144 |
0.150 |
0.095 |
0.036 |
0.037 |
0.036 |
0.020 |
|
|
2nd brand |
0.113 |
-1.061
|
0.069 |
0.030 |
0.030 |
0.029 |
0.017 |
||
|
Generic |
0.028 |
0.029 |
-0.345 |
0.007 |
0.006 |
0.007 |
0.017 |
||
Table 6: Mixed Logit Results for Beta Blockers
Model C: Normally-distributed coefficients for Generic and Angina constants
Model D: Normally-distributed coefficients for Generic and Angina constants &
normally-distributed error components of molecule specific constants
|
(C) |
(D) |
|||||
|
Variable name |
Estimate |
Standard error |
Estimate |
Standard error |
||
|
1 |
Price for HMO |
**-1.826 |
0.557 |
**-1.496 |
0.569 |
|
|
2 |
Price for Medicaid |
*-1.935 |
0.764 |
*-1.678 |
0.827 |
|
|
3 |
Price for Comm. Plan |
*-0.753 |
0.364 |
-0.383 |
0.425 |
|
|
4 |
Price for Self-paid |
-0.311 |
0.602 |
-0.065 |
0.616 |
|
|
5 |
log(Advertising cost) |
**0.527 |
0.040 |
**0.544 |
0.043 |
|
|
6 |
log(Age of product) |
**1.592 |
0.122 |
**1.618 |
0.136 |
|
|
7 |
Age * Generics |
-0.004 |
0.008 |
-0.004 |
0.008 |
|
|
8 |
Non-white * Generics |
-0.248 |
0.457 |
-0.258 |
0.530 |
|
|
9 |
Female * Generics |
-0.182 |
0.260 |
-0.181 |
0.302 |
|
|
10 |
Specialist * Generics |
-0.293 |
0.389 |
-0.413 |
0.417 |
|
|
11 |
Obesity * Angina |
-0.434 |
0.345 |
-0.404 |
0.346 |
|
|
12 |
Smoking * Angina |
0.843 |
0.560 |
0.804 |
0.547 |
|
|
13 |
Generic constant |
Mean |
*1.306 |
0.636 |
**1.565 |
0.606 |
|
14 |
Standard deviation |
**1.738 |
0.328 |
**1.651 |
0.332 |
|
|
15 |
Angina constant |
Mean |
**1.063 |
0.393 |
*0.836 |
0.352 |
|
16 |
Standard deviation |
**1.359 |
0.299 |
**1.045 |
0.288 |
|
|
17 |
Price adjustment for HMO |
**0.454 |
0.149 |
0.304 |
0.188 |
|
|
18 |
Price adjustment for Medicaid |
**1.041 |
0.212 |
**0.966 |
0.266 |
|
|
19 |
Price adjustment for Comm. Plan |
**0.470 |
0.084 |
**0.362 |
0.114 |
|
|
20 |
Price adjustment for Self-paid |
**0.896 |
0.151 |
**0.714 |
0.188 |
|
|
21 |
Molecule-specific constants |
Standard deviation |
**1.076 |
0.092 |
||
Number of observations 1426 1426
Log of likelihood function -2405.86 -2353.05
Adjusted likelihood ratio index 0.354 0.368
** 1% significant level
* 5% significant level
Table 7: Simulated Price Elasticities for Beta Blockers
|
HMO |
Own molecule |
Other molecules |
|||||||
|
Mono-poly |
Non-monopoly |
Mono-poly |
Non-monopoly |
||||||
|
Pioneer brand |
Pioneer brand |
2nd brand |
Generic |
Pioneer brand |
Pioneer brand |
2nd brand |
Generic |
||
|
Mono-poly |
Pioneer brand |
-1.494 |
0.024 |
0.008 |
0.008 |
0.006 |
|||
|
Non-mono-poly |
Pioneer brand |
-0.789 |
0.570 |
0.188 |
0.174 |
0.149 |
0.104 |
0.107 |
|
|
2nd brand |
0.001 |
-1.009
|
0.005 |
0.004 |
0.003 |
||||
|
Generic |
0.020 |
-0.565 |
0.012 |
0.009 |
0.010 |
0.024 |
|||
|
MEDICAID |
Own molecule |
Other molecules |
|||||||
|
Mono-poly |
Non-monopoly |
Mono-poly |
Non-monopoly |
||||||
|
Pioneer brand |
Pioneer brand |
2nd brand |
Generic |
Pioneer brand |
Pioneer brand |
2nd brand |
Generic |
||
|
Mono-poly |
Pioneer brand |
-1.664 |
0.021 |
0.007 |
0.007 |
0.005 |
|||
|
Non-mono-poly |
Pioneer brand |
-0.610 |
0.545 |
0.147 |
0.145 |
0.120 |
0.080 |
0.090 |
|
|
2nd brand |
0.008 |
-1.127 |
0.004 |
0.003 |
0.003 |
||||
|
Generic |
0.017 |
-0.646 |
0.010 |
0.007 |
0.009 |
0.021 |
|||
Table 7: Simulated Price Elasticities for Beta Blockers (Continued)
|
COMMERCIAL |
Own molecule |
Other molecules |
|||||||
|
Mono-poly |
Non-monopoly |
Mono-poly |
Non-monopoly |
||||||
|
Pioneer brand |
Pioneer brand |
2nd brand |
Generic |
Pioneer brand |
Pioneer brand |
2nd brand |
Generic |
||
|
Monopoly |
Pioneer brand |
-0.417 |
0.008 |
0.003 |
0.003 |
0.002 |
|||
|
Non-mono-poly |
Pioneer brand |
-0.111 |
0.138 |
0.004 |
0.024 |
0.021 |
0.007 |
0.016 |
|
|
2nd brand |
0.002 |
-0.269
|
0.001 |
0.001 |
0.001 |
||||
|
Generic |
0.004 |
-0.164 |
0.002 |
0.002 |
0.002 |
0.006 |
|||
|
SELF-PAID |
Own molecule |
Other molecules |
|||||||
|
Mono-poly |
Non-monopoly |
Mono-poly |
Non-monopoly |
||||||
|
Pioneer brand |
Pioneer brand |
2nd brand |
Generic |
Pioneer brand |
Pioneer brand |
2nd brand |
Generic |
||
|
Mono-poly |
Pioneer brand |
-0.072 |
0.001 |
0.0004 |
0.0004 |
0.0003 |
|||
|
Non-mono-poly |
Pioneer brand |
0.201 |
0.021 |
-0.145 |
-0.0514 |
0.0432 |
-0.059 |
-0.031 |
|
|
2nd brand |
0.0003 |
-0.046
|
0.0001 |
0.0001 |
0.0001 |
||||
|
Generic |
0.0005 |
-0.028 |
0.0003 |
0.0002 |
0.0002 |
0.001 |
|||
A Data Appendix
A.1 Antidepressants
A.1.1 Pharmacology of Antidepressants
There are several classes of drugs prescribed for the treatment of depression. These include Monoamine Oxidase Inhibitors (MAOIs), Tricyclics, Selective Serotonin Reuptake Inhibitors (SSRIs), and other structurally unrelated drugs. The efficacy rates of various classes of antidepressants are very similar to each other; hence, it appears that the degree of side effects carries more weight for differences in quality and attractiveness (aside from differences in individual reactions to each drug). The MAOIs and tricyclics are the oldest antidepressants, which emerged in the 1950s. Due to the severe risk of hypertensive crisis associated with the MAOIs, their usage today is very limited compared to other antidepressant drugs. Therefore, the MAOIs are excluded from my analysis.
There are twenty-eight antidepressant drugs in my analysis of data from 1991 to 1993. Potential side effects of these twenty-eight drugs are summarized in Table A.1. The treatment with tricyclic drugs is often accompanied by conditions such as drowsiness, weight gain, increased heart rate, decreased blood pressure, sexual dysfunction, etc. In addition, overdoses of tricyclics can be fatal. Aside from these side effects, tricyclic antidepressants have been successful in treating depression, with an efficacy rate of seventy percent. One should note that an individual patient’s responses to one tricyclic drug or another may vary, even though there is no significant efficacy rate difference among various tricyclics. Table A.1 also shows that SSRIs, introduced in the late 1980s, have significantly reduced side effects over tricyclics. Also, overdoses have much less severe outcomes.
The quality of drugs is characterized by many different attributes including the number of indications and the length of half-life as well as the type and frequency of side effects shown in Table A.1. It is a difficult task to construct a single quality indicator variable quantifying the quality of each product due to the multi-dimensional nature of "quality." To construct variables for each main aspect of quality and include them in regression models becomes increasingly similar to just using product-specific dummy variables, as the number of quality indicator variables increases. In my estimation, I use category-specific dummy variables in order to capture the effect of quality on demand.
Table A.2 summarizes marketing characteristics of antidepressant drugs from IMS data. The SSRIs have higher prices and marketing costs, because they are newer and have fewer side effects than other alternatives. All SSRIs are heavily marketed, as their annual marketing costs exceed twenty million dollars. Prozac was a breakthrough drug in the antidepressant category and has been very successful since its introduction in 1988. The success of Prozac motivated the entry of Zoloft, Paxil, and Effexor (launched in 1994) in subsequent years. Contrary to the large advertising costs of SSRIs, generic manufacturers appear to make no significant marketing efforts. Generic drugs which have been in the market for a while are often fifty percent cheaper than their brand name counterparts (e.g., Amitriptyline, Doxepin), though generics which entered the market more recently seem to have smaller discounts (e.g., Nortriptyline).
The patient and physician characteristics in the antidepressant market from the NAMCS data are summarized in Table A.3. Women are twice as likely to suffer from depression than men, and the NAMCS data show the same propensity in all insurance categories. The number of female patients found in the NAMCS data is about double the number of male patients. Also, the age distribution of patients in the NAMCS data agrees with the fact that depression is observed more commonly among the working age people of twenty to fifty years old.
The share of specialists in the NAMCS data exceeds fifty percent both in the commercial plan and self-paid sectors. Seeing a specialist tends to be more costly than seeing a primary care doctor. Therefore, it is expected that the share of specialists is higher for the commercial sector where coverage tends to be less restrictive, but is lower for the self-paid sector where patients have to cover the entire cost of office visit. The possible explanation is that patients in the self-paid sector may be sicker than in other sectors and have a greater need to see specialists. Due to the lack of coverage, people without insurance may wait longer for seeking treatment than people with insurance. This may be particularly true for the case of depression and other mental diseases, which are not necessarily manifested with physical pain and immediate health risk often associated with non-mental diseases.
A.2 Beta Blockers
A.2.1 Pharmacology of Beta Blockers
Figure A.1 illustrates a typical treatment algorithm defined by the Joint National Committee on Detection, Evaluation, and Treatment of High Blood Pressure. The report provides guidelines for hypertension therapies to physicians and other health professionals.
The first step in treating hypertensive patients is to encourage lifestyle modifications, such as weight reduction, tobacco avoidance, exercise, and restriction of sodium and alcohol. Drug therapy is often initiated after the failure of lifestyle modifications to control the blood pressure. There are several classes of anti-hypertensive drugs : diuretics, beta blockers, ACE inhibitors, calcium channel blockers, alpha-receptor blockers, and the alpha-beta blocker. For mild to moderate hypertension (often referred as stage 1 and stage 2 hypertension), the report recommends monotherapy (treatment with a single drug). Further, the report guides physicians to choose diuretics or beta blockers over other alternatives, for long-term controlled clinical trials have demonstrated the reduction of cardiovascular morbidity and mortality through the treatment with these agents. The alternative drugs - ACE inhibitors, calcium channel blockers, alpha-receptor blockers and the alpha-beta blocker - are encouraged to be reserved for special indications or when diuretics and beta blockers are found to be ineffective. For severe to very severe hypertension where control of blood pressure is not achieved with a single drug, physicians often increase the dose of the currently used medication and/or add a second or third drug from different classes.
Beta blockers are recommended for initial therapy as well as for the treatment of more severe hypertensive patients according to the guideline. All beta blocker agents are found to be fairly equivalent in terms of anti-hypertensive efficacy and adverse effects (The Merck Manual, 1992). The negative side effects of beta blockers include bronchospasm, insomnia, fatigue, lethargy, sexual dysfunction in men, exacerbation of heart failure, and depressed HDL (high-density lipoprotein) cholesterol. Since all beta blocker agents have similar adverse effects, the quality difference of various molecules is controlled by applying the dummy variable for molecules with indications for angina pectoris as well as hypertension in order to capture some advantages associated with multiple indications.
Marketing characteristics of beta blockers found in IMS data are summarized in Table A.4. Sixteen beta blocker agents which were indicated for the treatment of hypertension during 1991 to 1993. Atenolol, Metoprolol, Nadolol, and Propranolol have FDA approved indications for angina pectoris as well as hypertension. Similar to antidepressant data, generics have substantially smaller marketing costs compared to brand name drugs. The dates of generic entries in the beta blocker market are more recent than in the antidepressant market, ranging from 1985 to 1993.
Table A.5 characterizes the NAMCS data for beta blockers. According to the report of the joint national committee on treatment of hypertension, the prevalence of hypertension rises with age and is greater for blacks than for whites. Further, the report states that "in young adulthood and early middle age, high blood pressure prevalence is greater for men than for women; thereafter, the reverse is true" (pp.3). The age distribution of beta blocker users in the NAMCS data is skewed towards older ages in all four insurance categories, as expected. However, the share of black patients is not higher for the beta blocker market than for the overall markets in the NAMCS data. This outcome may be due to the fact that in general white patients are found to respond better to beta blockers, while black patients do better on diuretics and calcium channel blockers (The Merck Manual, 1992). Also, there are more female than male patients in all insurance categories. That is probably because the incidence of hypertension increases with age and at older ages women are more likely to suffer from high blood pressure.
The share of cardiologists in the commercial plan sector is around forty percent, which is much higher than all other categories where the share ranges from ten to thirty-three percent. Assuming that no one insurance category has a sicker pool of patients (e.g., the severity of patients’ illness in each insurance category is similar), this outcome confirms the notion that the restrictions of insurance coverage affect patients’ behavior. Only ten percent of patients in the self-paid sector saw a specialist as opposed to forty percent of patients in the commercial sector. Since seeing a specialist is much more expensive than a primary care doctor and commercial plans tend to be more generous and less restrictive than others, it is understandable that the commercial sector has the highest share of specialists.
Appendix Tables
Table A.1: Pharmacological Characteristics of Antidepressants
Tricyclics and Heterocyclics
|
Molecule Name |
FI * |
AC* |
DR* |
I/A* |
OH* |
CA* |
GID* |
WG* |
|
Amitriptyline (Elavil, Endep, Generics) |
2 |
4 |
4 |
0 |
4 |
3 |
0 |
4 |
|
Amoxapine (Asendin, Generics) |
1 |
2 |
2 |
2 |
2 |
3 |
0 |
1 |
|
Clomipramine (Anafranil) |
2 |
4 |
4 |
1 |
4 |
2 |
0 |
3 |
|
Desipramine (Norpramin, Pertofrane Generics) |
2 |
1 |
1 |
1 |
2 |
2 |
0 |
1 |
|
Doxepin (Sinequan, Generics) |
2 |
3 |
4 |
0 |
2 |
2 |
0 |
3 |
|
Imipramine (Tofranil, Janimine, Generics) |
2 |
3 |
3 |
1 |
4 |
3 |
1 |
3 |
|
Maprotiline (Ludiomil, Generics) |
1 |
3 |
3 |
1 |
2 |
3 |
3 |
2 |
|
Nortriptyline (Aventyl, Pamelor, Generics) |
2 |
1 |
2 |
0 |
2 |
2 |
0 |
1 |
|
Protriptyline (Vivactil) |
2 |
2 |
1 |
1 |
2 |
2 |
0 |
0 |
|
Trimipramine (Surmontil, Generics) |
2 |
3 |
4 |
0 |
4 |
2 |
0 |
3 |
Selective Serotonin Reuptake Inhibitors
|
Molecule Name |
FI |
AC |
DR |
I/A |
OH |
CA |
GID |
WG |
|
Fluoxetine (Prozac) |
1 |
0 |
0 |
2 |
0 |
0 |
3 |
0 |
|
Paroxetine (Paxil) |
1 |
0 |
0 |
2 |
0 |
0 |
3 |
0 |
|
Sertraline (Zoloft) |
1 |
0 |
0 |
2 |
0 |
0 |
3 |
0 |
Others
|
Molecule Name |
FI |
AC |
DR |
I/A |
OH |
CA |
GID |
WG |
|
Bupropion (Wellbutrin) |
.5 |
0 |
0 |
2 |
0 |
1 |
1 |
0 |
|
Trazodone (Desyrel, Generics) |
1 |
1 |
4 |
0 |
4 |
1 |
3 |
0 |
Table A.2: Marketing Characteristics of Antidepressants
Tricyclics and Heterocyclics
|
Molecule Name |
Product Name |
Brand |
Entry Date |
Average Price per Day (CPI adjusted) |
Avg. Annual Marketing Cost (in 1000, PPI adjusted) |
|
|
Amitriptyline |
Elavil |
Yes |
04/61 |
0.84 |
22 |
|
|
Endep |
Yes |
12/75 |
0.70 |
4 |
||
|
Amitriptyline |
No |
06/77 |
0.23 |
0 |
||
|
Amoxapine |
Asendin |
Yes |
10/80 |
2.31 |
67 |
|
|
Amoxapine |
No |
08/89 |
1.66 |
0 |
||
|
Clomipramine |
Anafranil |
Yes |
02/90 |
2.25 |
1932 |
|
|
Desipramine |
Norpramin |
Yes |
01/67 |
1.88 |
157 |
|
|
Pertofrane |
Yes |
12/71 |
2.35 |
3 |
||
|
Desipramine |
No |
07/87 |
0.85 |
18 |
||
|
Doxepin |
Sinequan |
Yes |
10/69 |
1.09 |
352 |
|
|
Doxepin |
No |
05/86 |
0.43 |
2 |
||
|
Imipramine |
Tofranil |
Yes |
10/59 |
1.33 |
42 |
|
|
Janimine |
Yes |
04/77 |
0.33 |
0 |
||
|
Imipramine |
No |
04/76 |
0.28 |
3 |
||
|
Maprotiline |
Ludiomil |
Yes |
01/81 |
1.20 |
20 |
|
|
Maprotiline |
No |
06/88 |
0.85 |
0 |
||
|
Nortriptyline |
Aventyl |
Yes |
12/64 |
1.65 |
3 |
|
|
Pamelor |
Yes |
09/77 |
1.57 |
4673 |
||
|
Nortriptyline |
No |
03/92 |
1.45 |
255 |
||
|
Protriptyline |
Vivactil |
Yes |
11/67 |
1.39 |
13 |
|
|
Trimipramine |
Surmontil |
Yes |
10/79 |
1.63 |
11 |
|
|
Trimipramine |
No |
01/88* |
1.01 |
0 |
||
* Trimipramine was discontinued in November 92.
Selective Serotonin Reuptake Inhibitors
|
Molecule Name |
Product Name |
Brand |
Entry Date |
Average Price per Day (CPI adjusted) |
Avg. Annual Marketing Cost (in 1000, PPI adjusted) |
|
Fluoxetine |
Prozac |
Yes |
01/88 |
2.38 |
29073 |
|
Paroxetine |
Paxil |
Yes |
01/93 |
1.87 |
56093 |
|
Sertraline |
Zoloft |
Yes |
02/92 |
2.18 |
41648 |
Others
|
Molecule Name |
Product Name |
Brand |
Entry Date |
Average Price per Day (CPI adjusted) |
Avg. Annual Marketing Cost (in 1000, PPI adjusted) |
|
Bupropion |
Wellbutrin |
Yes |
03/86 |
1.71 |
11960 |
|
Trazodone |
Desyrel |
Yes |
03/82 |
2.11 |
1961 |
|
Trazodone |
No |
10/86 |
0.66 |
6 |
Table A.3: Characteristics of Patients and Physicians in the Antidepressant Market
|
RX INSURANCE |
Frequency |
Percent |
|
HMO/Pre-paid |
517 |
15.22 % |
|
Medicaid |
358 |
10.54 % |
|
Commercial |
1878 |
55.28 % |
|
Self-paid |
644 |
18.96 % |
|
SEX |
HMO/Pre-paid |
Medicaid |
Commercial |
Self-paid |
|
Female |
67.12 % |
71.51 % |
66.61 % |
65.22 % |
|
Male |
32.88 % |
28.49 % |
33.39 % |
34.78 % |
|
RACE |
HMO/Pre-paid |
Medicaid |
Commercial |
Self-paid |
|
White |
89.94 % |
81.56 % |
95.15 % |
98.14 % |
|
Black |
8.12 % |
15.36 % |
3.67 % |
1.24 % |
|
Asian/Pacific Is. |
1.94 % |
2.23 % |
0.91 % |
0.62 % |
|
Indian/Eskimo |
0.00 % |
0.85 % |
0.27 % |
0.00 % |
|
AGE |
HMO/Pre-paid |
Medicaid |
Commercial |
Self-paid |
|
less than 12 |
2.90 % |
5.03 % |
1.17 % |
1.09 % |
|
12-19 |
6.38 % |
5.03 % |
3.30 % |
4.04 % |
|
20-29 |
10.06 % |
13.69 % |
9.64 % |
13.04 % |
|
30-39 |
24.18 % |
25.14 % |
22.58 % |
24.22 % |
|
40-49 |
30.37 % |
23.18 % |
28.17 % |
24.38 % |
|
50-59 |
12.38 % |
14.25 % |
17.52 % |
15.22 % |
|
60-69 |
7.35 % |
7.26 % |
8.57 % |
9.94 % |
|
70 or older |
6.38 % |
6.42 % |
9.05 % |
8.07 % |
|
SPECIALTY |
HMO/Pre-paid |
Medicaid |
Commercial |
Self-paid |
|
Non-Specialist |
50.29 % |
50.28 % |
41.32 % |
38.35 % |
|
Specialist * |
49.71 % |
49.72 % |
58.68 % |
61.65 % |
*
Psychiatry, Psychoanalysis, and Child Psychiatry.
Figure A.1: Treatment Algorithm for Hypertensive Patients
Lifestyle Modifications:
Weight reduction
Moderation of alcohol intake
Regular physical activity
Reduction of sodium intake
Smoking cessation
ß
Inadequate Response*
ß
Continue Lifestyle Modifications
Initial Pharmacological Selection:
ß
Inadequate Response*
ß
ß
Inadequate Response
*ß
Add a Second or Third Agent and/or Diuretic if Not Already Prescribed
Table A.4: Marketing Characteristics of Beta Blockers
Anti-hypertensive Agents
|
Molecule Name |
Product Name |
Brand |
Entry Date |
Average Price per Day (CPI adjusted) |
Avg. Annual Marketing Cost (in 1000, PPI adjusted) |
|
|
Acebutolol |
Sectral |
Yes |
12/84 |
1.29 |
1540 |
|
|
Betaxolol HCI |
Kerlone |
Yes |
10/89 |
0.84 |
1054 |
|
|
Carteolol HCI |
Cartrol |
Yes |
12/88 |
1.06 |
5 |
|
|
Penbutolol Sulfate |
Levatol |
Yes |
01/89 |
0.94 |
2567 |
|
|
Pindolol |
Visken |
Yes |
09/82 |
1.41 |
176 |
|
|
Pindolol |
No |
10/92 |
1.12 |
34 |
||
|
Timolol Maleate |
Blocadren |
Yes |
11/81 |
0.91 |
23 |
|
|
Timolol Maleate |
No |
04/89 |
0.59 |
1 |
||
Anti-hypertensive Agents with Indication for Angina
|
Molecule Name |
Product Name |
Brand |
Entry Date |
Average Price per Day (CPI adjusted) |
Avg. Annual Marketing Cost (in 1000, PPI adjusted) |
|||||
|
Atenolol |
Tenormin |
Yes |
08/81 |
0.93 |
16307 |
|||||
|
Atenolol |
No |
07/91 |
0.63 |
372 |
||||||
|
Metoprolol |
Lopressor |
Yes |
08/78 |
0.85 |
13218 |
|||||
|
Toprol XL |
Yes |
02/92 |
0.74 |
11441 |
||||||
|
Nadolol |
Corgard |
Yes |
12/79 |
1.09 |
4621 |
|||||
|
Nadolol |
No |
11/93 |
1.00 |
141 |
||||||
|
Propranolol HCI |
Inderal |
Yes |
11/67 |
0.98 |
1694 |
|||||
|
Propranolol HCI |
No |
07/85 |
0.37 |
21 |
||||||
Table A.5: Characteristics of Patients and Physicians in the Beta-Blocker Market
|
RX INSURANCE |
Frequency |
Percent |
|
HMO/Pre-paid |
256 |
17.95% |
|
Medicaid |
136 |
9.54% |
|
Commercial |
791 |
55.47% |
|
Self-paid |
243 |
17.04% |
|
SEX |
HMO/Pre-paid |
Medicaid |
Commercial |
Self-paid |
|
Female |
57.42 % |
63.24 % |
56.01 % |
59.26 % |
|
Male |
42.58 % |
36.76 % |
43.99 % |
40.74 % |
|
RACE |
HMO/Pre-paid |
Medicaid |
Commercial |
Self-paid |
|
White |
91.80 % |
79.41 % |
91.53 % |
92.59 % |
|
Black |
4.30 % |
16.91 % |
5.56 % |
6.17 % |
|
Asian/Pacific Is. |
3.90 % |
2.21 % |
2.78 % |
0.42 % |
|
Indian/Eskimo |
0.00 % |
1.47 % |
0.13 % |
0.82 % |
|
AGE |
HMO/Pre-paid |
Medicaid |
Commercial |
Self-paid |
|
less than 12 |
0.39 % |
1.47 % |
0.76 % |
0.00 % |
|
12-19 |
1.17 % |
1.47 % |
1.39 % |
1.65 % |
|
20-29 |
5.09 % |
5.08 % |
3.54 % |
4.53 % |
|
30-39 |
11.72 % |
5.47 % |
8.47 % |
9.47 % |
|
40-49 |
16.80 % |
7.03 % |
15.04 % |
18.52 % |
|
50-59 |
20.70 % |
16.91 % |
22.12 % |
24.69 % |
|
60-69 |
21.88 % |
13.28 % |
23.39 % |
23.46 % |
|
70 or older |
22.27 % |
22.06 % |
25.28 % |
17.70 % |
|
SPECIALTY |
HMO/Pre-paid |
Medicaid |
Commercial |
Self-paid |
|
Non-Specialist |
76.95 % |
75 % |
71.68 % |
90.53 % |
|
Specialist * |
23.05 % |
25 % |
28.32 % |
9.47 % |
*
Cardiologists.
Bibliography
American Society of Hospital Pharmacists (1993), American Hospital Formulary Service 1993.
Anderson, Simon P. and A. de Palma (1989), Discrete Choice Theory of Product Differentiation.
Anderson, Simon P. and A. de Palma (1992), "Multiproduct Firms: A Nested Logit Approach," Journal of Industrial Economics, XL (3), pp.261-76.
Ben-Akiva, Moshe and S. Lerman (1985), Discrete Choice Analysis, MIT Press, Cambridge, MA.
Berkow, Robert and A. Fletcher (1992), The Merck Manual, Sixteenth Edition, Merck Research Laboratories, Merck & Co., Inc., Rahway, NJ.
Berndt, Ernst R., I. Cockburn and Z. Griliches (1996), "Unmet Needs, Medical Discoveries, and Market Dynamics: Price Indexes for Anti-Depressant Drugs," mimeo.
Caves, Richard, M. Whinston, and M. Hurwitz (1991), "Patent Expiration, Entry, and Competition in the U.S. Pharmaceutical Industry," Brookings Papers on Economic Activity, Microeconomics, pp. 1-48.
CibaGeneva Pharmaceuticals(1995), Pharmacy Benefit Report - Trends and Forecasts 1995 Edition, New Jersey.
CibaGeneva Pharmaceuticals(1995), Pharmacy Benefit Report - 1995 Facts and Figures, New Jersey.
Dranove, David (1989), "Medicaid Drug Formulary Restrictions," Journal of Law & Economics Vol. XXXII, pp.143-162.
Ellison, Sara Fischer, I. Cockburn, Z. Griliches, and J. Hausman (1997), "Characteristics of Demand for Pharmaceutical Products: An Examination of Four Cephalosporins," RAND Journal of Economics, vol.28, No. 3, Autumn, pp.426-446.
Facts and Comparisons (1996), Inc., Drug Facts and Comparisons - loose-leaf drug information service, St. Louis, MO.
Food and Drug Administration (annual), Approved Drug Products with Therapeutic Equivalence Evaluations (a.k.a. the Orange Book), U.S. Department of Health and Human Services, Public Health Service, Center for Drug Evaluation and Research, Office of Management, Washington, D.C.
Frank, Richard and D. Salkever (1995), "Generic Entry and the Pricing of Pharmaceuticals," mimeo.
Frank, Richard and D. Salkever (1992), "Pricing, Patent Loss and the Market for Pharmaceutical," National Bureau of Economic Research Working Paper No. 3803.
Gondek, Kathleen, "Prescription Drug Payment Policy: Past, Present, and Future," Health Care Financing Review, vol. 15, number 3, pp.1-6.
Grabowski, Henry and J. Vernon (1992), "Brand Loyalty, Entry and Price Competition in Pharmaceutical after the 1984 Drug Act," Journal of Law and Economics.
Greene, William H. (1990), Econometric Analysis, Maxwell Macmillan International Publishing Group, NY.
Hellerstein, Judith (1994), "The Demand for Post-Patent Prescription Pharmaceuticals," National Bureau of Economic Research Working Paper No.4981.
IMS America (1991-1993), National Prescription Audit, National Journal Audit, and Office Promotion Report, Plymouth Meeting, PA.
Kozma, Christopher M., R. Schulz, M. Dickson, J. Dye, E. Rox, D. Holdford, L. Michael, W. Yates Jr. and T. Young (1993), "Economic Impact of Cost-Containment Strategies in Third Party Programmes in the US (Part II)," PharmacoEconomics, 4(3), pp. 187-202.
Liang, Bryan A. (1996), "The Anticompetitive Nature of Brand-name Firm Introduction of Generics Before Patent Expiration," The Antitrust Bulletin, Fall, pp. 599-635.
Masson, Alison and R. Steiner (1985), Generic Substitution and Prescription Drug Prices: Economic Effects of State Drug Product Selection Laws, Federal Trade Commission, Washington, D.C.
McGahan, Anita Am. (1994), "Industry Structure and Competitive Advantage: an Outsider’s Perspective on the Changes in Prescription Pharmaceuticals," Harvard Business Review, v72, n6, pp.115-125.
Merck & Co. (1993), Managed Care: Health Maintenance Organizations/Preferred Provider Organizations, West Point, PA.
Merck & Co. (1992), The Merck Manual of Diagnosis and Therapy Sixteenth Edition, Rahway, NJ.
Moore, William J. and R. Newman (1993), "Drug Formulary Restrictions as a Cost-Containment Policy in Medicaid Programs," Journal of Law & Economics, vol. XXXVI, pp.71-97.
Morton, Fiona Scott (1994), "Barriers to Entry, Brand Advertising, and Generic Entry in the U.S. Pharmaceutical Industry," MIT dissertation.
Pharmaceutical Manufacturers Association (1991), Facts as a Glance, 1991 Edition.
Reeder, Eugene C., E. Lingle, R. Schulz, R. Mauch Jr., B. Nightengale, C. Pedersen, M. Watrous and S. Zetzl (1993), "Economic Impact of Cost-Containment Strategies in Third Party Programmes in the US (Part I)," Pharmaaco Economics, 4(2), pp. 92-103.
Spratto, George and A.Woods (1996), Delmar’s Drug Guide for Nurses 1996, Delmar Publishers.
Standard & Poor (1993, 1994, 1995, 1996), Standard & Poor’s Industry Surveys.
Stern, Scott (1994), "Product Demand in Pharmaceutical Markets," Stanford University dissertation.
Train, Kenneth (1986), Qualitative Choice Analysis: Theory, Econometrics, and an Application to Automobile Demand, MIT Press, Cambridge, MA.
Revelt, David and K. Train (1996), "Incentives for Appliance Efficiency: Random-Parameters Logit Models of Households’ Choices," working paper, Department of Economics, University of California, Berkeley.
U.S. Department of Health and Human Services (1994), Approved Drug Products with Therapeutic Equivalence Evaluations (a.k.a. FDA Orange Book), Washington, D.C.
US Department of Health and Human Services, Health Care Financing Administration (1993), MEDICAID spDATA System, Characteristics of Medicaid State Programs, Washington, D.C.
US Department of Health and Human Services, Health Care Financing Administration (1992), Report to Congress, Medicaid and Institutions for Mental Diseases, Washington, D.C.
Wagner, Judith (1994), "Long-Term Care and Drug Benefits Under Health Care Reform," Office of Technology Assessment, Washington, D.C.
Wagner, Judith (1993), "Pharmaceutical Marketplace Reform: Is Competition the Right Prescription?" Office of Technology Assessment, Washington, D.C.
Wiggins, Steven and R. Maness (1994), "Price Competition in Pharmaceutical Markets," mimeo.